Different composition of the human and the mouse gammadelta T cell receptor explains different phenotypes of CD3gamma and CD3delta immunodeficiencies
- PMID: 17923503
- PMCID: PMC2118495
- DOI: 10.1084/jem.20070782
Different composition of the human and the mouse gammadelta T cell receptor explains different phenotypes of CD3gamma and CD3delta immunodeficiencies
Erratum in
- J Exp Med. 2007 Nov 26;204(12):3049
Abstract
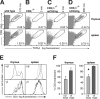
The gammadelta T cell receptor for antigen (TCR) comprises the clonotypic TCRgammadelta, the CD3 (CD3gammaepsilon and/or CD3deltaepsilon), and the zetazeta dimers. gammadelta T cells do not develop in CD3gamma-deficient mice, whereas human patients lacking CD3gamma have abundant peripheral blood gammadelta T cells expressing high gammadelta TCR levels. In an attempt to identify the molecular basis for these discordant phenotypes, we determined the stoichiometries of mouse and human gammadelta TCRs using blue native polyacrylamide gel electrophoresis and anti-TCR-specific antibodies. The gammadelta TCR isolated in digitonin from primary and cultured human gammadelta T cells includes CD3delta, with a TCRgammadeltaCD3epsilon(2)deltagammazeta(2) stoichiometry. In CD3gamma-deficient patients, this may allow substitution of CD3gamma by the CD3delta chain and thereby support gammadelta T cell development. In contrast, the mouse gammadelta TCR does not incorporate CD3delta and has a TCRgammadeltaCD3epsilon(2)gamma(2)zeta(2) stoichiometry. CD3gamma-deficient mice exhibit a block in gammadelta T cell development. A human, but not a mouse, CD3delta transgene rescues gammadelta T cell development in mice lacking both mouse CD3delta and CD3gamma chains. This suggests important structural and/or functional differences between human and mouse CD3delta chains during gammadelta T cell development. Collectively, our results indicate that the different gammadelta T cell phenotypes between CD3gamma-deficient humans and mice can be explained by differences in their gammadelta TCR composition.
Figures



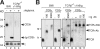
Similar articles
-
Human CD3γ, but not CD3δ, haploinsufficiency differentially impairs γδ versus αβ surface TCR expression.BMC Immunol. 2013 Jan 21;14:3. doi: 10.1186/1471-2172-14-3. BMC Immunol. 2013. PMID: 23336327 Free PMC article.
-
Essential and partially overlapping role of CD3gamma and CD3delta for development of alphabeta and gammadelta T lymphocytes.J Exp Med. 1998 Oct 5;188(7):1375-80. doi: 10.1084/jem.188.7.1375. J Exp Med. 1998. PMID: 9763617 Free PMC article.
-
On the role of CD3delta chains in TCRgammadelta/CD3 complexes during assembly and membrane expression.Scand J Immunol. 2001 Jul-Aug;54(1-2):155-62. doi: 10.1046/j.1365-3083.2001.00938.x. Scand J Immunol. 2001. PMID: 11439162
-
[Expression anomalies of the CD3-TCR complex expression and immunodeficiencies].Med Sci (Paris). 2007 Feb;23(2):161-6. doi: 10.1051/medsci/2007232161. Med Sci (Paris). 2007. PMID: 17291425 Review. French.
-
An architectural perspective on signaling by the pre-, alphabeta and gammadelta T cell receptors.Immunol Rev. 2003 Feb;191:28-37. doi: 10.1034/j.1600-065x.2003.00011.x. Immunol Rev. 2003. PMID: 12614349 Review.
Cited by
-
Structure of the chicken CD3εδ/γ heterodimer and its assembly with the αβT cell receptor.J Biol Chem. 2014 Mar 21;289(12):8240-51. doi: 10.1074/jbc.M113.544965. Epub 2014 Jan 31. J Biol Chem. 2014. PMID: 24488493 Free PMC article.
-
Gamma delta T cells and their immunotherapeutic potential in cancer.Biomark Res. 2025 Mar 28;13(1):51. doi: 10.1186/s40364-025-00762-6. Biomark Res. 2025. PMID: 40148988 Free PMC article. Review.
-
Five Layers of Receptor Signaling in γδ T-Cell Differentiation and Activation.Front Immunol. 2015 Jan 26;6:15. doi: 10.3389/fimmu.2015.00015. eCollection 2015. Front Immunol. 2015. PMID: 25674089 Free PMC article. Review.
-
T Cell Microvilli: Finger-Shaped External Structures Linked to the Fate of T Cells.Immune Netw. 2023 Feb 21;23(1):e3. doi: 10.4110/in.2023.23.e3. eCollection 2023 Feb. Immune Netw. 2023. PMID: 36911802 Free PMC article. Review.
-
Rapid cloning, expression, and functional characterization of paired αβ and γδ T-cell receptor chains from single-cell analysis.Mol Ther Methods Clin Dev. 2016 Jan 27;3:15054. doi: 10.1038/mtm.2015.54. eCollection 2016. Mol Ther Methods Clin Dev. 2016. PMID: 26858965 Free PMC article.
References
-
- Recio, M.J., M.A. Moreno-Pelayo, S.S. Kilic, A.C. Guardo, O. Sanal, L.M. Allende, V. Perez-Flores, A. Mencia, S. Modamio-Hoybjor, E. Seoane, and J.R. Regueiro. 2007. Differential biological role of CD3 chains revealed by human immunodeficiencies. J. Immunol. 178:2556–2564. - PubMed
-
- Dadi, H.K., A.J. Simon, and C.M. Roifman. 2003. Effect of CD3δ deficiency on maturation of αβ and γδ T-cell lineages in severe combined immunodeficieny. N. Engl. J. Med. 349:1821–1828. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

