Phase II study of low-dose paclitaxel and cisplatin as a second-line therapy after 5-fluorouracil/platinum chemotherapy in gastric cancer
- PMID: 17923737
- PMCID: PMC2694374
- DOI: 10.3346/jkms.2007.22.S.S115
Phase II study of low-dose paclitaxel and cisplatin as a second-line therapy after 5-fluorouracil/platinum chemotherapy in gastric cancer
Abstract
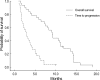
This study was performed to evaluate the efficacy and toxicity of low-dose paclitaxel/cisplatin chemotherapy in patients with metastatic or recurrent gastric cancer that had failed 5-fluorouracil/platinum-based chemotherapy. Thirty-two patients with documented progression on or within 6 months after discontinuing 5-fluorouracil/platinum-based chemotherapy were enrolled. As a second-line treatment, paclitaxel (145 mg/m(2)) and cisplatin (60 mg/m(2)) was administered on day 1 every 3 weeks. Among 32 patients enrolled, 8 (25%) responded partially to paclitaxel/cisplatin, 8 (25%) had stable disease, and 14 (44%) had progressive disease. Two patients (6%) were not evaluable. The median time to progression (TTP) and overall survival for all patients were 2.9 months and 9.1 months, respectively. The most common hematologic toxicity was anemia (47%). Grade 3 neutropenia developed in three patients (9%), but no other grade 3/4 hematologic toxicity occurred. The most common non-hematologic toxicities were emesis (31%) and peripheral neuropathy (38%). Three cases (9%) of grade 3/4 emesis and 2 cases (6%) of grade 3 peripheral neuropathy developed. In conclusion, low-dose paclitaxel and cisplatin chemotherapy showed moderate activity with favorable toxicity profiles. However, relatively short TTP of this regimen warrants the development of more effective paclitaxel-based regimens other than combination with cisplatin in these patients as second-line therapies.
Figures
References
-
- Shin HR, Ahn YO, Bae JM, Shin MH, Lee DH, Lee CW, Ohrr HC, Ahn DH, Ferlay J, Parkin DM, Oh DK, Park JG. Cancer incidence in Korea. Cancer Res Treat. 2002;34:405–408. - PubMed
-
- Murad AM, Santiago FF, Petroianu A, Rocha PR, Rodrigues MA, Rausch M. Modified therapy with 5-fluorouracil, doxorubicin, and methotrexate in advanced gastric cancer. Cancer. 1993;72:37–41. - PubMed
-
- Glimelius B, Ekstrom K, Hoffman K, Graf W, Sjoden PO, Haglund U, Svensson C, Enander LK, Linne T, Sellstrom H, Heuman R. Randomized comparison between chemotherapy plus best supportive care with best supportive care in advanced gastric cancer. Ann Oncol. 1997;8:163–168. - PubMed
-
- Kim NK, Park YS, Heo DS, Suh C, Kim SY, Park KC, Kang YK, Shin DB, Kim HT, Kim HJ, Kang WK, Suh CI, Bang YJ. A phase III randomized study of 5-fluorouracil and cisplatin versus 5-fluorouracil, doxorubicin, and mitomycin C versus 5-fluorouracil alone in the treatment of advanced gastric cancer. Cancer. 1993;71:3813–3818. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical
Research Materials