Herbivore-induced terpenoid emission in Medicago truncatula: concerted action of jasmonate, ethylene and calcium signaling
- PMID: 17924138
- PMCID: PMC2756395
- DOI: 10.1007/s00425-007-0631-y
Herbivore-induced terpenoid emission in Medicago truncatula: concerted action of jasmonate, ethylene and calcium signaling
Abstract
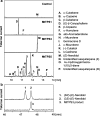
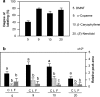
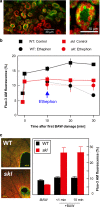
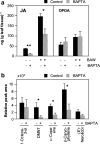
Plant volatiles emitted by Medicago truncatula in response to feeding larvae of Spodoptera exigua are composed of a complex blend of terpenoids. The cDNAs of three terpene synthases (TPSs), which contribute to the blend of terpenoids, were cloned from M. truncatula. Their functional characterization proved MtTPS1 to be a beta-caryophyllene synthase and MtTPS5 to be a multi-product sesquiterpene synthase. MtTPS3 encodes a bifunctional enzyme producing (E)-nerolidol and geranyllinalool (precursors of C11 and C16 homoterpenes) from different prenyl diphosphates serving as substrates. The addition of jasmonic acid (JA) induced expression of the TPS genes, but terpenoid emission was higher from plants treated with JA and the ethylene precursor 1-amino-cyclopropyl-1-carboxylic acid. Compared to infested wild-type M. truncatula plants, lower amounts of various sesquiterpenes and a C11-homoterpene were released from an ethylene-insensitive mutant skl. This difference coincided with lower transcript levels of MtTPS5 and of 1-deoxy-D: -xylulose-5-phosphate synthase (MtDXS2) in the damaged skl leaves. Moreover, ethephon, an ethylene-releasing compound, modified the extent and mode of the herbivore-stimulated Ca2+ variations in the cytoplasm that is necessary for both JA and terpene biosynthesis. Thus, ethylene contributes to the herbivory-induced terpenoid biosynthesis at least twice: by modulating both early signaling events such as cytoplasmic Ca2+-influx and the downstream JA-dependent biosynthesis of terpenoids.
Figures


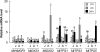
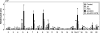
References
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1105/tpc.016253', 'is_inner': False, 'url': 'https://doi.org/10.1105/tpc.016253'}, {'type': 'PMC', 'value': 'PMC282818', 'is_inner': False, 'url': 'https://pmc.ncbi.nlm.nih.gov/articles/PMC282818/'}, {'type': 'PubMed', 'value': '14630967', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/14630967/'}]}
- Aharoni A, Giri AP, Deuerlein S, Griepink F, de Kogel WJ, Verstappen FWA, Verhoeven HA, Jongsma MA, Schwab W, Bouwmeester HJ (2003) Terpenoid metabolism in wild-type and transgenic Arabidopsis plants. Plant Cell 15:2866–2884 - PMC - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1016/j.tplants.2005.10.005', 'is_inner': False, 'url': 'https://doi.org/10.1016/j.tplants.2005.10.005'}, {'type': 'PubMed', 'value': '16290212', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/16290212/'}]}
- Aharoni A, Jongsma MA, Bouwmeester HJ (2005) Volatile science? Metabolic engineering of terpenoids in plants. Trends Plant Sci 10:594–602 - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1007/s00425-006-0301-5', 'is_inner': False, 'url': 'https://doi.org/10.1007/s00425-006-0301-5'}, {'type': 'PubMed', 'value': '16786318', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/16786318/'}]}
- Ament K, Van Schie CC, Bouwmeester HJ, Haring MA, Schuurink RC (2006) Induction of a leaf specific geranylgeranyl pyrophosphate synthase and emission of (E,E)-4,8,12-trimethyltrideca-1,3,7,11-tetraene in tomato are dependent on both jasmonic acid and salicylic acid signaling pathways. Planta 224:1197–1208 - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1038/35020072', 'is_inner': False, 'url': 'https://doi.org/10.1038/35020072'}, {'type': 'PubMed', 'value': '10952311', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/10952311/'}]}
- Arimura G, Ozawa R, Shimoda T, Nishioka T, Boland W, Takabayashi J (2000) Herbivory-induced volatiles elicit defence genes in lima bean leaves. Nature 406:512–515 - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1111/j.1365-313X.2003.01987.x', 'is_inner': False, 'url': 'https://doi.org/10.1111/j.1365-313x.2003.01987.x'}, {'type': 'PubMed', 'value': '14756770', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/14756770/'}]}
- Arimura G, Huber DPW, Bohlmann J (2004a) Forest tent caterpillars (Malacosoma disstria) induce local and systemic diurnal emissions of terpenoid volatiles in hybrid poplar (Populus trichocarpa × deltoides): cDNA cloning, functional characterization, and patterns of gene expression of (-)-germacrene D synthase, PtdTPS1. Plant J 37:603–616 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

