The exchange activities of [Fe] hydrogenase (iron-sulfur-cluster-free hydrogenase) from methanogenic archaea in comparison with the exchange activities of [FeFe] and [NiFe] hydrogenases
- PMID: 17924153
- PMCID: PMC2757585
- DOI: 10.1007/s00775-007-0302-2
The exchange activities of [Fe] hydrogenase (iron-sulfur-cluster-free hydrogenase) from methanogenic archaea in comparison with the exchange activities of [FeFe] and [NiFe] hydrogenases
Abstract
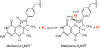
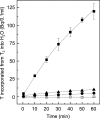
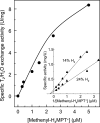
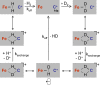
[Fe] hydrogenase (iron-sulfur-cluster-free hydrogenase) catalyzes the reversible reduction of methenyltetrahydromethanopterin (methenyl-H4MPT+) with H2 to methylene-H4MPT, a reaction involved in methanogenesis from H2 and CO2 in many methanogenic archaea. The enzyme harbors an iron-containing cofactor, in which a low-spin iron is complexed by a pyridone, two CO and a cysteine sulfur. [Fe] hydrogenase is thus similar to [NiFe] and [FeFe] hydrogenases, in which a low-spin iron carbonyl complex, albeit in a dinuclear metal center, is also involved in H2 activation. Like the [NiFe] and [FeFe] hydrogenases, [Fe] hydrogenase catalyzes an active exchange of H2 with protons of water; however, this activity is dependent on the presence of the hydride-accepting methenyl-H4MPT+. In its absence the exchange activity is only 0.01% of that in its presence. The residual activity has been attributed to the presence of traces of methenyl-H4MPT+ in the enzyme preparations, but it could also reflect a weak binding of H2 to the iron in the absence of methenyl-H4MPT+. To test this we reinvestigated the exchange activity with [Fe] hydrogenase reconstituted from apoprotein heterologously produced in Escherichia coli and highly purified iron-containing cofactor and found that in the absence of added methenyl-H4MPT+ the exchange activity was below the detection limit of the tritium method employed (0.1 nmol min(-1) mg(-1)). The finding reiterates that for H2 activation by [Fe] hydrogenase the presence of the hydride-accepting methenyl-H4MPT+ is essentially required. This differentiates [Fe] hydrogenase from [FeFe] and [NiFe] hydrogenases, which actively catalyze H2/H2O exchange in the absence of exogenous electron acceptors.
Figures


Similar articles
-
Preparation of [Fe]-hydrogenase from methanogenic archaea.Methods Enzymol. 2011;494:119-37. doi: 10.1016/B978-0-12-385112-3.00007-X. Methods Enzymol. 2011. PMID: 21402213
-
Hydrogenases from methanogenic archaea, nickel, a novel cofactor, and H2 storage.Annu Rev Biochem. 2010;79:507-36. doi: 10.1146/annurev.biochem.030508.152103. Annu Rev Biochem. 2010. PMID: 20235826 Review.
-
Scaffold-Based Functional Models of [Fe]-Hydrogenase (Hmd): Building the Bridge between Biological Structure and Molecular Function.Acc Chem Res. 2020 Aug 18;53(8):1637-1647. doi: 10.1021/acs.accounts.0c00315. Epub 2020 Aug 4. Acc Chem Res. 2020. PMID: 32786339
-
Hydrogenases and H(+)-reduction in primary energy conservation.Results Probl Cell Differ. 2008;45:223-52. doi: 10.1007/400_2006_027. Results Probl Cell Differ. 2008. PMID: 18500479 Review.
-
Towards a functional identification of catalytically inactive [Fe]-hydrogenase paralogs.FEBS J. 2015 Sep;282(17):3412-23. doi: 10.1111/febs.13351. Epub 2015 Jul 14. FEBS J. 2015. PMID: 26094576
Cited by
-
Reversible oxygen-tolerant hydrogenase carried by free-living N2-fixing bacteria isolated from the rhizospheres of rice, maize, and wheat.Microbiologyopen. 2012 Dec;1(4):349-61. doi: 10.1002/mbo3.37. Epub 2012 Sep 12. Microbiologyopen. 2012. PMID: 23233392 Free PMC article.
-
Amidine dications: isolation and [Fe]-hydrogenase-related hydrogenation.J Am Chem Soc. 2009 Jul 8;131(26):9174-5. doi: 10.1021/ja9035847. J Am Chem Soc. 2009. PMID: 19534467 Free PMC article.
-
Amidine dications as superelectrophiles.J Am Chem Soc. 2009 Dec 16;131(49):17980-5. doi: 10.1021/ja908191k. J Am Chem Soc. 2009. PMID: 19916499 Free PMC article.
-
Radical S-adenosyl-L-methionine chemistry in the synthesis of hydrogenase and nitrogenase metal cofactors.J Biol Chem. 2015 Feb 13;290(7):3987-94. doi: 10.1074/jbc.R114.578161. Epub 2014 Dec 4. J Biol Chem. 2015. PMID: 25477518 Free PMC article. Review.
-
Nitrogen fixation and hydrogen metabolism in cyanobacteria.Microbiol Mol Biol Rev. 2010 Dec;74(4):529-51. doi: 10.1128/MMBR.00033-10. Microbiol Mol Biol Rev. 2010. PMID: 21119016 Free PMC article. Review.
References
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'PubMed', 'value': '11524134', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/11524134/'}]}
- Vignais PM, Billoud B, Meyer J (2001) FEMS Microbiol Rev 25:455–501 - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1016/j.ccr.2005.01.026', 'is_inner': False, 'url': 'https://doi.org/10.1016/j.ccr.2005.01.026'}]}
- Vignais PM (2005) Coord Chem Rev 249:1677–1690
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1098/rsta.2004.1528', 'is_inner': False, 'url': 'https://doi.org/10.1098/rsta.2004.1528'}, {'type': 'PubMed', 'value': '15991402', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/15991402/'}]}
- Armstrong FA, Albracht SP (2005) Philos Trans A Math Phys Eng Sci 363:937–954; discussion 1035–1040 - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1002/tcr.20111', 'is_inner': False, 'url': 'https://doi.org/10.1002/tcr.20111'}, {'type': 'PubMed', 'value': '17304591', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/17304591/'}]}
- Shima S, Thauer RK (2007) Chem Rec 7:37–46 - PubMed
-
- Volbeda A, Fontecilla-Camps JC (2003) Dalton Trans 4030–4038 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

