Sugar binding induces an outward facing conformation of LacY
- PMID: 17925435
- PMCID: PMC2034228
- DOI: 10.1073/pnas.0708258104
Sugar binding induces an outward facing conformation of LacY
Abstract
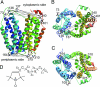
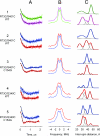
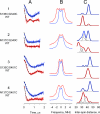
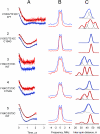
According to x-ray structure, the lactose permease (LacY) is a monomer organized into N- and C-terminal six-helix bundles that form a deep internal cavity open on the cytoplasmic side with a single sugar-binding site at the apex. The periplasmic side of the molecule is closed. During sugar/H(+) symport, a cavity facing the periplasmic side is thought to open with closure of the inward-facing cytoplasmic cavity so that the sugar-binding site is alternately accessible to either face of the membrane. Double electron-electron resonance (DEER) is used here to measure interhelical distance changes induced by sugar binding to LacY. Nitroxide-labeled paired-Cys replacements were constructed at the ends of transmembrane helices on the cytoplasmic or periplasmic sides of wild-type LacY and in the conformationally restricted mutant Cys-154-->Gly. Distances were then determined in the presence of galactosidic or nongalactosidic sugars. Strikingly, specific binding causes conformational rearrangement on both sides of the molecule. On the cytoplasmic side, each of six nitroxide-labeled pairs exhibits decreased interspin distances ranging from 4 to 21 A. Conversely, on the periplasmic side, each of three spin-labeled pairs shows increased distances ranging from 4 to 14 A. Thus, the inward-facing cytoplasmic cavity closes, and a cleft opens on the tightly packed periplasmic side. In the Cys-154-->Gly mutant, sugar-induced closing is observed on the cytoplasmic face, but little or no change occurs on periplasmic side. The DEER measurements in conjunction with molecular modeling based on the x-ray structure provide strong support for the alternative access model and reveal a structure for the outward-facing conformer of LacY.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Residues gating the periplasmic pathway of LacY.J Mol Biol. 2009 Nov 27;394(2):219-25. doi: 10.1016/j.jmb.2009.09.043. Epub 2009 Sep 23. J Mol Biol. 2009. PMID: 19781551 Free PMC article.
-
The Cys154-->Gly mutation in LacY causes constitutive opening of the hydrophilic periplasmic pathway.J Mol Biol. 2008 Jun 13;379(4):695-703. doi: 10.1016/j.jmb.2008.04.015. Epub 2008 Apr 11. J Mol Biol. 2008. PMID: 18485365 Free PMC article.
-
Opening the periplasmic cavity in lactose permease is the limiting step for sugar binding.Proc Natl Acad Sci U S A. 2011 Sep 13;108(37):15147-51. doi: 10.1073/pnas.1112157108. Epub 2011 Sep 6. Proc Natl Acad Sci U S A. 2011. PMID: 21896727 Free PMC article.
-
The alternating access transport mechanism in LacY.J Membr Biol. 2011 Jan;239(1-2):85-93. doi: 10.1007/s00232-010-9327-5. Epub 2010 Dec 16. J Membr Biol. 2011. PMID: 21161516 Free PMC article. Review.
-
It takes two to tango: The dance of the permease.J Gen Physiol. 2019 Jul 1;151(7):878-886. doi: 10.1085/jgp.201912377. Epub 2019 May 30. J Gen Physiol. 2019. PMID: 31147449 Free PMC article. Review.
Cited by
-
Apo-intermediate in the transport cycle of lactose permease (LacY).Proc Natl Acad Sci U S A. 2012 Oct 30;109(44):E2970-8. doi: 10.1073/pnas.1211183109. Epub 2012 Sep 24. Proc Natl Acad Sci U S A. 2012. PMID: 23012238 Free PMC article.
-
YidC assists the stepwise and stochastic folding of membrane proteins.Nat Chem Biol. 2016 Nov;12(11):911-917. doi: 10.1038/nchembio.2169. Epub 2016 Sep 5. Nat Chem Biol. 2016. PMID: 27595331 Free PMC article.
-
Single residue within the antigen translocation complex TAP controls the epitope repertoire by stabilizing a receptive conformation.Proc Natl Acad Sci U S A. 2010 May 18;107(20):9135-40. doi: 10.1073/pnas.1001308107. Epub 2010 May 3. Proc Natl Acad Sci U S A. 2010. PMID: 20439763 Free PMC article.
-
Conformational cycle and ion-coupling mechanism of the Na+/hydantoin transporter Mhp1.Proc Natl Acad Sci U S A. 2014 Oct 14;111(41):14752-7. doi: 10.1073/pnas.1410431111. Epub 2014 Sep 29. Proc Natl Acad Sci U S A. 2014. PMID: 25267652 Free PMC article.
-
Probing of the rates of alternating access in LacY with Trp fluorescence.Proc Natl Acad Sci U S A. 2009 Dec 22;106(51):21561-6. doi: 10.1073/pnas.0911434106. Epub 2009 Dec 3. Proc Natl Acad Sci U S A. 2009. PMID: 19959662 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
- R56 DK051131/DK/NIDDK NIH HHS/United States
- R01 DK051131/DK/NIDDK NIH HHS/United States
- R01 EY005216/EY/NEI NIH HHS/United States
- GM074929/GM/NIGMS NIH HHS/United States
- R37 EY005216/EY/NEI NIH HHS/United States
- U54 GM074929/GM/NIGMS NIH HHS/United States
- EY05216/EY/NEI NIH HHS/United States
- GM073210/GM/NIGMS NIH HHS/United States
- DK069463/DK/NIDDK NIH HHS/United States
- DK051131/DK/NIDDK NIH HHS/United States
- P50 GM073210/GM/NIGMS NIH HHS/United States
- R56 DK069463/DK/NIDDK NIH HHS/United States
- R01 DK069463/DK/NIDDK NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

