Flight initiation and maintenance deficits in flies with genetically altered biogenic amine levels
- PMID: 17928454
- PMCID: PMC6672854
- DOI: 10.1523/JNEUROSCI.2704-07.2007
Flight initiation and maintenance deficits in flies with genetically altered biogenic amine levels
Abstract
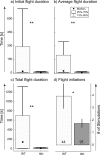
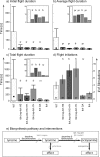
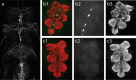
Insect flight is one of the fastest, most intense and most energy-demanding motor behaviors. It is modulated on multiple levels by the biogenic amine octopamine. Within the CNS, octopamine acts directly on the flight central pattern generator, and it affects motivational states. In the periphery, octopamine sensitizes sensory receptors, alters muscle contraction kinetics, and enhances flight muscle glycolysis. This study addresses the roles for octopamine and its precursor tyramine in flight behavior by genetic and pharmacological manipulation in Drosophila. Octopamine is not the natural signal for flight initiation because flies lacking octopamine [tyramine-beta-hydroxylase (TbetaH) null mutants] can fly. However, they show profound differences with respect to flight initiation and flight maintenance compared with wild-type controls. The morphology, kinematics, and development of the flight machinery are not impaired in TbetaH mutants because wing-beat frequencies and amplitudes, flight muscle structure, and overall dendritic structure of flight motoneurons are unaffected in TbetaH mutants. Accordingly, the flight behavior phenotypes can be rescued acutely in adult flies. Flight deficits are rescued by substituting octopamine but also by blocking the receptors for tyramine, which is enriched in TbetaH mutants. Conversely, ablating all neurons containing octopamine or tyramine phenocopies TbetaH mutants. Therefore, both octopamine and tyramine systems are simultaneously involved in regulating flight initiation and maintenance. Different sets of rescue experiments indicate different sites of action for both amines. These findings are consistent with a complex system of multiple amines orchestrating the control of motor behaviors on multiple levels rather than single amines eliciting single behaviors.
Figures



Similar articles
-
Coordination and modulation of locomotion pattern generators in Drosophila larvae: effects of altered biogenic amine levels by the tyramine beta hydroxlyase mutation.J Neurosci. 2006 Feb 1;26(5):1486-98. doi: 10.1523/JNEUROSCI.4749-05.2006. J Neurosci. 2006. PMID: 16452672 Free PMC article.
-
Trace amines differentially regulate adult locomotor activity, cocaine sensitivity, and female fertility in Drosophila melanogaster.Dev Neurobiol. 2007 Sep 1;67(10):1396-405. doi: 10.1002/dneu.20459. Dev Neurobiol. 2007. PMID: 17638385
-
Sensitivity to expression levels underlies differential dominance of a putative null allele of the Drosophila tβh gene in behavioral phenotypes.PLoS Biol. 2021 May 10;19(5):e3001228. doi: 10.1371/journal.pbio.3001228. eCollection 2021 May. PLoS Biol. 2021. PMID: 33970909 Free PMC article.
-
Tyramine and octopamine: ruling behavior and metabolism.Annu Rev Entomol. 2005;50:447-77. doi: 10.1146/annurev.ento.50.071803.130404. Annu Rev Entomol. 2005. PMID: 15355245 Review.
-
Molecular and pharmacological properties of insect biogenic amine receptors: lessons from Drosophila melanogaster and Apis mellifera.Arch Insect Biochem Physiol. 2001 Sep;48(1):13-38. doi: 10.1002/arch.1055. Arch Insect Biochem Physiol. 2001. PMID: 11519073 Review.
Cited by
-
Short-term memories in Drosophila are governed by general and specific genetic systems.Learn Mem. 2010 Apr 23;17(5):246-51. doi: 10.1101/lm.1706110. Print 2010 May. Learn Mem. 2010. PMID: 20418404 Free PMC article. Review.
-
Neuroarchitecture of aminergic systems in the larval ventral ganglion of Drosophila melanogaster.PLoS One. 2008 Mar 26;3(3):e1848. doi: 10.1371/journal.pone.0001848. PLoS One. 2008. PMID: 18365004 Free PMC article.
-
Dendrites are dispensable for basic motoneuron function but essential for fine tuning of behavior.Proc Natl Acad Sci U S A. 2014 Dec 16;111(50):18049-54. doi: 10.1073/pnas.1416247111. Epub 2014 Dec 1. Proc Natl Acad Sci U S A. 2014. PMID: 25453076 Free PMC article.
-
Octopaminergic neurons have multiple targets in Drosophila larval mushroom body calyx and can modulate behavioral odor discrimination.Learn Mem. 2021 Jan 15;28(2):53-71. doi: 10.1101/lm.052159.120. Print 2021 Feb. Learn Mem. 2021. PMID: 33452115 Free PMC article.
-
Report on the 12th symposium on invertebrate neurobiology held 31 August-4 September 2011 at the Balaton Limnological Research Institute of the Hungarian Academy of Sciences, Tihany, Hungary.Invert Neurosci. 2012 Jun;12(1):69-79. doi: 10.1007/s10158-012-0131-z. Epub 2012 Apr 6. Invert Neurosci. 2012. PMID: 22481213
References
-
- Alkema MJ, Hunter-Ensor M, Ringstad N, Horvitz HR. Tyramine functions independently of octopamine in the Caenorhabditis elegans nervous system. Neuron. 2005;46:247–260. - PubMed
-
- Arakawa S, Gocayne JD, McCombie WR, Urquhart DA, Hall LM, Fraser CM, Venter JC. Cloning, localization, and permanent expression of a Drosophila tyramine receptor. Neuron. 1990;2:342–354. - PubMed
-
- Baier A, Wittek B, Brembs B. Drosophila as a model organism for the neurobiology of aggression. J Exp Biol. 2002;205:1233–1240. - PubMed
-
- Blenau W, Baumann A. Aminergic signal transduction in invertebrates: focus on tyramine and octopamine receptors. Recent Res Dev Neurochem. 2003;6:225–240.
-
- Blumenthal EM. Regulation of chloride permeability by endogenously produced tyramine in the Drosophila malphigian tubule. Am J Cell Physiol. 2003;284:C718–C728. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
