Role of the Trypanosoma brucei natural cysteine peptidase inhibitor ICP in differentiation and virulence
- PMID: 17944830
- PMCID: PMC2680270
- DOI: 10.1111/j.1365-2958.2007.05970.x
Role of the Trypanosoma brucei natural cysteine peptidase inhibitor ICP in differentiation and virulence
Abstract
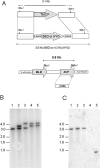
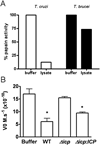
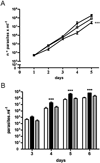
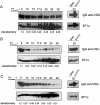
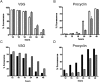
ICP is a chagasin-family natural tight binding inhibitor of Clan CA, family C1 cysteine peptidases (CPs). We investigated the role of ICP in Trypanosoma brucei by generating bloodstream form ICP-deficient mutants (Deltaicp). A threefold increase in CP activity was detected in lysates of Deltaicp, which was restored to the levels in wild type parasites by re-expression of the gene in the null mutant. Deltaicp displayed slower growth in culture and increased resistance to a trypanocidal synthetic CP inhibitor. More efficient exchange of the variant surface glycoprotein (VSG) to procyclin during differentiation from bloodstream to procyclic form was observed in Deltaicp, a phenotype that was reversed in the presence of synthetic CP inhibitors. Furthermore, we showed that degradation of anti-VSG IgG is abolished when parasites are pretreated with synthetic CP inhibitors, and that parasites lacking ICP degrade IgG more efficiently than wild type. In addition, Deltaicp reached higher parasitemia than wild type parasites in infected mice, suggesting that ICP modulates parasite infectivity. Taken together, these data suggest that CPs of T. brucei bloodstream form play a role in surface coat exchange during differentiation, in the degradation of internalized IgG and in parasite infectivity, and that their function is regulated by ICP.
Figures

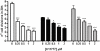
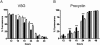
Similar articles
-
Trypanosoma brucei rhodesiense Inhibitor of Cysteine Peptidase (ICP) Is Required for Virulence in Mice and to Attenuate the Inflammatory Response.Int J Mol Sci. 2022 Dec 30;24(1):656. doi: 10.3390/ijms24010656. Int J Mol Sci. 2022. PMID: 36614101 Free PMC article.
-
Differentiation of a culture-adapted mutant bloodstream form of Trypanosoma brucei into the procyclic form results in growth arrest of the cells.Mol Biochem Parasitol. 1995 Jun;72(1-2):215-25. doi: 10.1016/0166-6851(95)00081-b. Mol Biochem Parasitol. 1995. PMID: 8538691
-
The mRNA cap methyltransferase gene TbCMT1 is not essential in vitro but is a virulence factor in vivo for bloodstream form Trypanosoma brucei.PLoS One. 2018 Jul 24;13(7):e0201263. doi: 10.1371/journal.pone.0201263. eCollection 2018. PLoS One. 2018. PMID: 30040830 Free PMC article.
-
Regulation of antigen gene expression in Trypanosoma brucei.Trends Parasitol. 2005 Nov;21(11):517-20. doi: 10.1016/j.pt.2005.08.016. Epub 2005 Aug 29. Trends Parasitol. 2005. PMID: 16126458 Review.
-
Update on relevant trypanosome peptidases: Validated targets and future challenges.Biochim Biophys Acta Proteins Proteom. 2021 Feb;1869(2):140577. doi: 10.1016/j.bbapap.2020.140577. Epub 2020 Nov 30. Biochim Biophys Acta Proteins Proteom. 2021. PMID: 33271348 Review.
Cited by
-
Chalcogen Derivatives for the Treatment of African Trypanosomiasis: Biological Evaluation of Thio- and Seleno-Semicarbazones and Their Azole Derivatives.ACS Omega. 2025 Jun 5;10(23):24872-24886. doi: 10.1021/acsomega.5c02014. eCollection 2025 Jun 17. ACS Omega. 2025. PMID: 40547650 Free PMC article.
-
The cathepsin L of Toxoplasma gondii (TgCPL) and its endogenous macromolecular inhibitor, toxostatin.Mol Biochem Parasitol. 2009 Mar;164(1):86-94. doi: 10.1016/j.molbiopara.2008.11.012. Epub 2008 Dec 6. Mol Biochem Parasitol. 2009. PMID: 19111576 Free PMC article.
-
Trypanosoma brucei rhodesiense Inhibitor of Cysteine Peptidase (ICP) Is Required for Virulence in Mice and to Attenuate the Inflammatory Response.Int J Mol Sci. 2022 Dec 30;24(1):656. doi: 10.3390/ijms24010656. Int J Mol Sci. 2022. PMID: 36614101 Free PMC article.
-
Exploring the activity of the putative Δ6-desaturase and its role in bloodstream form life-cycle transitions in Trypanosoma brucei.PLoS Pathog. 2025 Feb 18;21(2):e1012691. doi: 10.1371/journal.ppat.1012691. eCollection 2025 Feb. PLoS Pathog. 2025. PMID: 39965027 Free PMC article.
-
Decoding the network of Trypanosoma brucei proteins that determines sensitivity to apolipoprotein-L1.PLoS Pathog. 2018 Jan 18;14(1):e1006855. doi: 10.1371/journal.ppat.1006855. eCollection 2018 Jan. PLoS Pathog. 2018. PMID: 29346416 Free PMC article.
References
-
- Abrahamson M, Alvarez-Fernandez M, Nathanson CM. Cystatins. Biochem Soc Symp. 2003;70:179–199. - PubMed
-
- Berriman M, Ghedin E, Hertz-Fowler C, Blandin G, Renauld H, Bartholomeu DC, et al. The genome of the African trypanosome Trypanosoma brucei. Science. 2005;309:416–422. - PubMed
-
- Caffrey CR, Hansell E, Lucas KD, Brinen LS, Hernandez AA, Cheng JN, et al. Active site mapping, biochemical properties and subcellular localization of rhodesain, the major cysteine protease of Trypanosoma Brucei Rhodesiense. Mol Biochem Parasitol. 2001;118:61–73. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

