Mussel-inspired surface chemistry for multifunctional coatings
- PMID: 17947576
- PMCID: PMC2601629
- DOI: 10.1126/science.1147241
Mussel-inspired surface chemistry for multifunctional coatings
Abstract
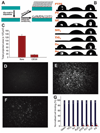
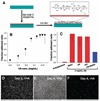
We report a method to form multifunctional polymer coatings through simple dip-coating of objects in an aqueous solution of dopamine. Inspired by the composition of adhesive proteins in mussels, we used dopamine self-polymerization to form thin, surface-adherent polydopamine films onto a wide range of inorganic and organic materials, including noble metals, oxides, polymers, semiconductors, and ceramics. Secondary reactions can be used to create a variety of ad-layers, including self-assembled monolayers through deposition of long-chain molecular building blocks, metal films by electroless metallization, and bioinert and bioactive surfaces via grafting of macromolecules.
Figures


Comment in
-
Bioinspired Coordination Micelles Integrating High Stability, Triggered Cargo Release, and Magnetic Resonance Imaging.ACS Appl Mater Interfaces. 2017 Jan 11;9(1):80-91. doi: 10.1021/acsami.6b09425. Epub 2016 Dec 21. ACS Appl Mater Interfaces. 2017. PMID: 27957858
Similar articles
-
Dopamine-Supported Metallization of Polyolefins─A Contribution to Transfer to an Eco-friendly and Efficient Technological Process.ACS Appl Mater Interfaces. 2022 Feb 2;14(4):5921-5931. doi: 10.1021/acsami.1c19575. Epub 2022 Jan 18. ACS Appl Mater Interfaces. 2022. PMID: 35040627
-
Nanostructured polymeric coatings based on chitosan and dopamine-modified hyaluronic acid for biomedical applications.Small. 2014 Jun 25;10(12):2459-69. doi: 10.1002/smll.201303568. Epub 2014 Mar 10. Small. 2014. PMID: 24616168
-
Formation, Removal, and Reformation of Surface Coatings on Various Metal Oxide Surfaces Inspired by Mussel Adhesives.ACS Appl Mater Interfaces. 2015 Nov 11;7(44):24656-62. doi: 10.1021/acsami.5b06910. Epub 2015 Oct 30. ACS Appl Mater Interfaces. 2015. PMID: 26492561
-
Materials from Mussel-Inspired Chemistry for Cell and Tissue Engineering Applications.Biomacromolecules. 2015 Sep 14;16(9):2541-55. doi: 10.1021/acs.biomac.5b00852. Epub 2015 Aug 25. Biomacromolecules. 2015. PMID: 26280621 Review.
-
Biomedical and Clinical Importance of Mussel-Inspired Polymers and Materials.Mar Drugs. 2015 Nov 11;13(11):6792-817. doi: 10.3390/md13116792. Mar Drugs. 2015. PMID: 26569266 Free PMC article. Review.
Cited by
-
Li[Li0.2Ni 0.16Mn 0.56Co 0.08]O 2 Nanoparticle/Carbon Composite Using Polydopamine Binding Agent for Enhanced Electrochemical Performance.Nanoscale Res Lett. 2015 Dec;10(1):986. doi: 10.1186/s11671-015-0986-0. Epub 2015 Jun 26. Nanoscale Res Lett. 2015. PMID: 26111979 Free PMC article.
-
Self-Assembly and Genetically Engineered Hydrogels.Adv Biochem Eng Biotechnol. 2021;178:169-196. doi: 10.1007/10_2021_165. Adv Biochem Eng Biotechnol. 2021. PMID: 33851233 Review.
-
Evaluation of Degree of Conversion, Resin-Dentin Bond Strength, and Durability of Polydopamine Incorporated Total Etch Adhesive System.Front Dent. 2020 Dec 17;17:35. doi: 10.18502/fid.v17i35.5199. eCollection 2020. Front Dent. 2020. PMID: 36042804 Free PMC article.
-
Establishment of a protocol for determining gastrointestinal transit time in mice using barium and radiopaque markers.Korean J Radiol. 2013 Jan-Feb;14(1):45-50. doi: 10.3348/kjr.2013.14.1.45. Epub 2012 Dec 28. Korean J Radiol. 2013. PMID: 23323030 Free PMC article.
-
Zn-contained mussel-inspired film on Mg alloy for inhibiting bacterial infection and promoting bone regeneration.Regen Biomater. 2020 Sep 30;8(1):rbaa044. doi: 10.1093/rb/rbaa044. eCollection 2021 Feb 1. Regen Biomater. 2020. PMID: 33732490 Free PMC article.
References
-
- Ratner BD, Hoffman AS, editors. Biomaterials Science: An Introduction to Materials in Medicine. ed. 2. San Diego, CA: Elsevier Academic; 2004.
-
- Ahn J-H, et al. Science. 2006;314:1754. - PubMed
-
- Alivisatos P. Nat. Biotechnol. 2004;22:47. - PubMed
-
- Langer R. Science. 2001;293:58. - PubMed
-
- Love JC, Estroff LA, Kriebel JK, Nuzzo RG, Whitesides GM. Chem. Rev. 2005;105:1103. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

