High glucose levels upregulate upstream stimulatory factor 2 gene transcription in mesangial cells
- PMID: 17955499
- PMCID: PMC9084927
- DOI: 10.1002/jcb.21585
High glucose levels upregulate upstream stimulatory factor 2 gene transcription in mesangial cells
Abstract
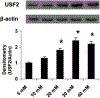
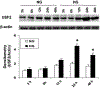
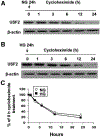
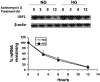
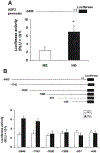
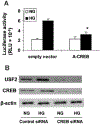
Previously, we demonstrated that upstream stimulatory factor 2 (USF2) mediates high glucose-induced thrombospondin1 (TSP1) gene expression and TGF-beta activity in glomerular mesangial cells and plays a role in diabetic renal complications. In the present studies, we further determined the molecular mechanisms by which high glucose levels regulate USF2 gene expression. In primary rat mesangial cells, we found that glucose treatment time and dose-dependently up-regulated USF2 expression (mRNA and protein). By using cycloheximide to block the de novo protein synthesis, similar rate of USF2 degradation was found under either normal glucose or high glucose conditions. USF2 mRNA stability was not altered by high glucose treatment. Furthermore, high glucose treatment stimulated USF2 gene promoter activity. By using the luciferase-promoter deletion assay, site-directed mutagenesis, and transactivation assay, we identified a glucose-responsive element in the USF2 gene promoter (-1,740 to -1,620, relative to the transcription start site) and demonstrated that glucose-induced USF2 expression is mediated through a cAMP-response element-binding protein (CREB)-dependent transactivation of the USF2 promoter. Furthermore, siRNA-mediated CREB knock down abolished glucose-induced USF2 expression. Taken together, these data indicate that high glucose levels up-regulate USF2 gene transcription in mesangial cells through CREB-dependent transactivation of the USF2 promoter.
Figures
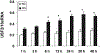


Similar articles
-
Glycated albumin upregulates upstream stimulatory factor 2 gene transcription in mesangial cells.Am J Physiol Renal Physiol. 2010 Jul;299(1):F121-7. doi: 10.1152/ajprenal.00074.2010. Epub 2010 Apr 21. Am J Physiol Renal Physiol. 2010. PMID: 20410211 Free PMC article.
-
Glucose up-regulates thrombospondin 1 gene transcription and transforming growth factor-beta activity through antagonism of cGMP-dependent protein kinase repression via upstream stimulatory factor 2.J Biol Chem. 2004 Aug 13;279(33):34311-22. doi: 10.1074/jbc.M401629200. Epub 2004 Jun 7. J Biol Chem. 2004. PMID: 15184388
-
High glucose upregulates upstream stimulatory factor 2 in human renal proximal tubular cells through angiotensin II-dependent activation of CREB.Nephron Exp Nephrol. 2011;117(3):e62-70. doi: 10.1159/000320593. Epub 2010 Sep 1. Nephron Exp Nephrol. 2011. PMID: 20814220 Free PMC article.
-
Activation of renal renin-angiotensin system in upstream stimulatory factor 2 transgenic mice.Am J Physiol Renal Physiol. 2009 Feb;296(2):F257-65. doi: 10.1152/ajprenal.90493.2008. Epub 2008 Nov 12. Am J Physiol Renal Physiol. 2009. PMID: 19004931
-
Overexpression of upstream stimulatory factor 2 accelerates diabetic kidney injury.Am J Physiol Renal Physiol. 2007 Nov;293(5):F1727-35. doi: 10.1152/ajprenal.00316.2007. Epub 2007 Sep 19. Am J Physiol Renal Physiol. 2007. PMID: 17881461
Cited by
-
Multiple E-boxes in the distal promoter of the rat pyruvate carboxylase gene function as a glucose-responsive element.PLoS One. 2014 Jul 23;9(7):e102730. doi: 10.1371/journal.pone.0102730. eCollection 2014. PLoS One. 2014. PMID: 25054881 Free PMC article.
-
Cell surface GRP78 regulates TGFβ1-mediated profibrotic responses via TSP1 in diabetic kidney disease.Front Pharmacol. 2023 Feb 24;14:1098321. doi: 10.3389/fphar.2023.1098321. eCollection 2023. Front Pharmacol. 2023. PMID: 36909183 Free PMC article.
-
Integrative single-cell characterization of a frugivorous and an insectivorous bat kidney and pancreas.Nat Commun. 2024 Jan 9;15(1):12. doi: 10.1038/s41467-023-44186-y. Nat Commun. 2024. PMID: 38195585 Free PMC article.
-
Transcriptional Regulation Factors of the Human Mitochondrial Aspartate/Glutamate Carrier Gene, Isoform 2 (SLC25A13): USF1 as Basal Factor and FOXA2 as Activator in Liver Cells.Int J Mol Sci. 2019 Apr 16;20(8):1888. doi: 10.3390/ijms20081888. Int J Mol Sci. 2019. PMID: 30995827 Free PMC article.
-
Blocking TSP1 Ameliorates Diabetes Mellitus-Induced Erectile Dysfunction by Inhibiting the TGF-β/SMAD Pathway.World J Mens Health. 2025 Jul;43(3):580-594. doi: 10.5534/wjmh.240065. Epub 2024 Aug 14. World J Mens Health. 2025. PMID: 39344111 Free PMC article.
References
-
- Bidder M, Shao JS, Charlton-Kachigian N, Loewy AP, Semenkovich CF, Towler DA. 2002. Osteopontin transcription in aortic vascular smooth muscle cells is controlled by glucose-regulated upstream stimulatory factor and activator protein-1 activities. J Biol Chem 277: 44485–44496. - PubMed
-
- Bloomgarden ZT. 2005. Diabetic nephropathy. Diabetes Care 28:745–751. - PubMed
-
- Corre S, Galibert MD. 2005. Upstream stimulating factors: Highly versatile stress-responsive transcription factors. Pigment Cell Res 18:337–348. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

