High speed two-photon imaging of calcium dynamics in dendritic spines: consequences for spine calcium kinetics and buffer capacity
- PMID: 17957255
- PMCID: PMC2034355
- DOI: 10.1371/journal.pone.0001073
High speed two-photon imaging of calcium dynamics in dendritic spines: consequences for spine calcium kinetics and buffer capacity
Abstract
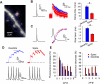
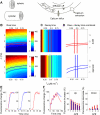
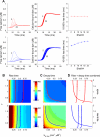
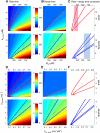
Rapid calcium concentration changes in postsynaptic structures are crucial for synaptic plasticity. Thus far, the determinants of postsynaptic calcium dynamics have been studied predominantly based on the decay kinetics of calcium transients. Calcium rise times in spines in response to single action potentials (AP) are almost never measured due to technical limitations, but they could be crucial for synaptic plasticity. With high-speed, precisely-targeted, two-photon point imaging we measured both calcium rise and decay kinetics in spines and secondary dendrites in neocortical pyramidal neurons. We found that both rise and decay kinetics of changes in calcium-indicator fluorescence are about twice as fast in spines. During AP trains, spine calcium changes follow each AP, but not in dendrites. Apart from the higher surface-to-volume ratio (SVR), we observed that neocortical dendritic spines have a markedly smaller endogenous buffer capacity with respect to their parental dendrites. Calcium influx time course and calcium extrusion rate were both in the same range for spines and dendrites when fitted with a dynamic multi-compartment model that included calcium binding kinetics and diffusion. In a subsequent analysis we used this model to investigate which parameters are critical determinants in spine calcium dynamics. The model confirmed the experimental findings: a higher SVR is not sufficient by itself to explain the faster rise time kinetics in spines, but only when paired with a lower buffer capacity in spines. Simulations at zero calcium-dye conditions show that calmodulin is more efficiently activated in spines, which indicates that spine morphology and buffering conditions in neocortical spines favor synaptic plasticity.
Conflict of interest statement
Figures

Similar articles
-
Mechanisms of calcium decay kinetics in hippocampal spines: role of spine calcium pumps and calcium diffusion through the spine neck in biochemical compartmentalization.J Neurosci. 2000 Mar 1;20(5):1722-34. doi: 10.1523/JNEUROSCI.20-05-01722.2000. J Neurosci. 2000. PMID: 10684874 Free PMC article.
-
Calcium signalling in medial intercalated cell dendrites and spines.J Physiol. 2017 Aug 15;595(16):5653-5669. doi: 10.1113/JP274261. Epub 2017 Jul 16. J Physiol. 2017. PMID: 28594440 Free PMC article.
-
Diffusion in a dendritic spine: the role of geometry.Phys Rev E Stat Nonlin Soft Matter Phys. 2007 Aug;76(2 Pt 1):021922. doi: 10.1103/PhysRevE.76.021922. Epub 2007 Aug 21. Phys Rev E Stat Nonlin Soft Matter Phys. 2007. PMID: 17930080
-
Calcium dynamics in spines: link to synaptic plasticity.Exp Physiol. 2002 Nov;87(6):725-31. doi: 10.1113/eph8702463. Exp Physiol. 2002. PMID: 12530404 Review.
-
From form to function: calcium compartmentalization in dendritic spines.Nat Neurosci. 2000 Jul;3(7):653-9. doi: 10.1038/76609. Nat Neurosci. 2000. PMID: 10862697 Review.
Cited by
-
Buffer mobility and the regulation of neuronal calcium domains.Front Cell Neurosci. 2015 Feb 20;9:48. doi: 10.3389/fncel.2015.00048. eCollection 2015. Front Cell Neurosci. 2015. PMID: 25750615 Free PMC article. Review.
-
Label-free live brain imaging and targeted patching with third-harmonic generation microscopy.Proc Natl Acad Sci U S A. 2011 Apr 12;108(15):5970-5. doi: 10.1073/pnas.1018743108. Epub 2011 Mar 28. Proc Natl Acad Sci U S A. 2011. PMID: 21444784 Free PMC article.
-
The diffusional properties of dendrites depend on the density of dendritic spines.Eur J Neurosci. 2011 Aug;34(4):561-8. doi: 10.1111/j.1460-9568.2011.07785.x. Epub 2011 Jul 19. Eur J Neurosci. 2011. PMID: 21771115 Free PMC article.
-
Spike inference from calcium imaging using sequential Monte Carlo methods.Biophys J. 2009 Jul 22;97(2):636-55. doi: 10.1016/j.bpj.2008.08.005. Biophys J. 2009. PMID: 19619479 Free PMC article.
-
Activity-dependent gating of calcium spikes by A-type K+ channels controls climbing fiber signaling in Purkinje cell dendrites.Neuron. 2014 Oct 1;84(1):137-151. doi: 10.1016/j.neuron.2014.08.035. Epub 2014 Sep 11. Neuron. 2014. PMID: 25220810 Free PMC article.
References
-
- Yuste R, Denk W. Dendritic spines as basic functional units of neuronal integration. Nature. 1995;375:682–684. - PubMed
-
- Yuste R, Majewska A, Holthoff K. From form to function: calcium compartmentalization in dendritic spines. NatNeurosci. 2000;3:653–659. - PubMed
-
- Sabatini BL, Maravall M, Svoboda K. Ca(2+) signaling in dendritic spines. Curr Opin Neurobiol. 2001;11:349–356. - PubMed
-
- Zucker RS. Calcium- and activity-dependent synaptic plasticity. Curr Opin Neurobiol. 1999;9:305–313. - PubMed
-
- Yang SN, Tang YG, Zucker RS. Selective induction of LTP and LTD by postsynaptic [Ca2+]i elevation. J Neurophysiol. 1999;81:781–787. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

