Ternary borate-nucleoside complex stabilization by ribonuclease A demonstrates phosphate mimicry
- PMID: 17957392
- PMCID: PMC2888155
- DOI: 10.1007/s00775-007-0311-1
Ternary borate-nucleoside complex stabilization by ribonuclease A demonstrates phosphate mimicry
Abstract
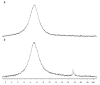
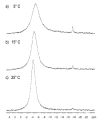
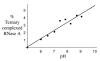
Phosphate esters play a central role in cellular energetics, biochemical activation, signal transduction and conformational switching. The structural homology of the borate anion with phosphate, combined with its ability to spontaneously esterify hydroxyl groups, suggested that phosphate ester recognition sites on proteins might exhibit significant affinity for nonenzymatically formed borate esters. (11)B NMR studies and activity measurements on ribonuclease A (RNase A) in the presence of borate and several cytidine analogs demonstrate the formation of a stable ternary RNase A.3'-deoxycytidine-2'-borate ternary complex that mimics the complex formed between RNase A and a 2'-cytidine monophosphate (2'-CMP) inhibitor. Alternatively, no slowly exchanging borate resonance is observed for a ternary RNase A, borate, 2'-deoxycytidine mixture, demonstrating the critical importance of the 2'-hydroxyl group for complex formation. Titration of the ternary complex with 2'-CMP shows that it can displace the bound borate ester with a binding constant that is close to the reported inhibition constant of RNase A by 2'-CMP. RNase A binding of a cyclic cytidine-2',3'-borate ester, which is a structural homolog of the cytidine-2',3'-cyclic phosphate substrate, could also be demonstrated. The apparent dissociation constant for the cytidine-2',3'-borate.RNase A complex is 0.8 mM, which compares with a Michaelis constant of 11 mM for cytidine-2',3'-cyclic phosphate at pH 7, indicating considerably stronger binding. However, the value is 1,000-fold larger than the reported dissociation constant of the RNase A complex with uridine-vanadate. These results are consistent with recent reports suggesting that in situ formation of borate esters that mimic the corresponding phosphate esters supports enzyme catalysis.
Figures








Similar articles
-
The crystal structure of ribonuclease A in complex with thymidine-3'-monophosphate provides further insight into ligand binding.Proteins. 2010 Aug 15;78(11):2459-68. doi: 10.1002/prot.22754. Proteins. 2010. PMID: 20602460
-
NMR and crystallographic characterization of adventitious borate binding by trypsin.Bioconjug Chem. 2006 Mar-Apr;17(2):300-8. doi: 10.1021/bc0502210. Bioconjug Chem. 2006. PMID: 16536459
-
Coulombic effects of remote subsites on the active site of ribonuclease A.Biochemistry. 1998 Dec 15;37(50):17386-401. doi: 10.1021/bi981369s. Biochemistry. 1998. PMID: 9860854
-
[Structures and functions of ribonucleases].Yakugaku Zasshi. 1997 Sep;117(9):561-82. doi: 10.1248/yakushi1947.117.9_561. Yakugaku Zasshi. 1997. PMID: 9357326 Review. Japanese.
-
Crystal Chemistry of High-Temperature Borates.Molecules. 2020 May 25;25(10):2450. doi: 10.3390/molecules25102450. Molecules. 2020. PMID: 32466152 Free PMC article. Review.
Cited by
-
Is boron a prebiotic element? A mini-review of the essentiality of boron for the appearance of life on earth.Orig Life Evol Biosph. 2012 Feb;42(1):3-17. doi: 10.1007/s11084-012-9269-2. Epub 2012 Apr 17. Orig Life Evol Biosph. 2012. PMID: 22528885 Review.
-
Synthesis of Boron-Containing Nucleoside Analogs.J Org Chem. 2024 Feb 2;89(3):1556-1566. doi: 10.1021/acs.joc.3c02179. Epub 2024 Jan 16. J Org Chem. 2024. PMID: 38227951 Free PMC article.
References
-
- Gresser MJ. J. Biol Chem. 1981;256:5981–5983. - PubMed
-
- Moore SA, Moennich DMC, Gresser MJ. J. Biol. Chem. 1983;258:6266–6271. - PubMed
-
- Borah B, Chen CW, Egan W, Miller M, Wlodawer A, Cohen JS. Biochemistry. 1985;24:2058–2067. - PubMed
-
- Rehder D, Holst H, Quaas R, Hinrichs W, Hahn U, Saenger W. J. Inorg Biochem. 1989;37:141–150. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

