Protein kinase Cdelta and calmodulin regulate epidermal growth factor receptor recycling from early endosomes through Arp2/3 complex and cortactin
- PMID: 17959830
- PMCID: PMC2174165
- DOI: 10.1091/mbc.e07-05-0411
Protein kinase Cdelta and calmodulin regulate epidermal growth factor receptor recycling from early endosomes through Arp2/3 complex and cortactin
Abstract
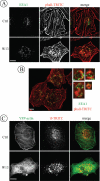
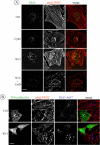
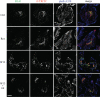
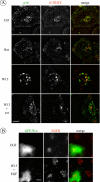
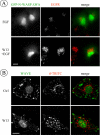
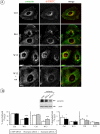
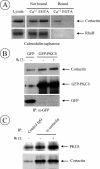
The intracellular trafficking of the epidermal growth factor receptor (EGFR) is regulated by a cross-talk between calmodulin (CaM) and protein kinase Cdelta (PKCdelta). On inhibition of CaM, PKCdelta promotes the formation of enlarged early endosomes and blocks EGFR recycling and degradation. Here, we show that PKCdelta impairs EGFR trafficking due to the formation of an F-actin coat surrounding early endosomes. The PKCdelta-induced polymerization of actin is orchestrated by the Arp2/3 complex and requires the interaction of cortactin with PKCdelta. Accordingly, inhibition of actin polymerization by using cytochalasin D or by overexpression of active cofilin, restored the normal morphology of the organelle and the recycling of EGFR. Similar results were obtained after down-regulation of cortactin and the sequestration of the Arp2/3 complex. Furthermore we demonstrate an interaction of cortactin with CaM and PKCdelta, the latter being dependent on CaM inhibition. In summary, this study provides the first evidence that CaM and PKCdelta organize actin dynamics in the early endosomal compartment, thereby regulating the intracellular trafficking of EGFR.
Figures

Similar articles
-
PI(3,5)P2 controls endosomal branched actin dynamics by regulating cortactin-actin interactions.J Cell Biol. 2015 Aug 31;210(5):753-69. doi: 10.1083/jcb.201412127. J Cell Biol. 2015. PMID: 26323691 Free PMC article.
-
Cortactin regulates cofilin and N-WASp activities to control the stages of invadopodium assembly and maturation.J Cell Biol. 2009 Aug 24;186(4):571-87. doi: 10.1083/jcb.200812176. J Cell Biol. 2009. PMID: 19704022 Free PMC article.
-
A Cortactin-CD2-associated protein (CD2AP) complex provides a novel link between epidermal growth factor receptor endocytosis and the actin cytoskeleton.J Biol Chem. 2003 Jun 13;278(24):21805-13. doi: 10.1074/jbc.M211407200. Epub 2003 Apr 2. J Biol Chem. 2003. PMID: 12672817
-
Actin-dependent endosomal receptor recycling.Curr Opin Cell Biol. 2019 Feb;56:22-33. doi: 10.1016/j.ceb.2018.08.006. Epub 2018 Sep 15. Curr Opin Cell Biol. 2019. PMID: 30227382 Review.
-
Cellular and pathophysiological consequences of Arp2/3 complex inhibition: role of inhibitory proteins and pharmacological compounds.Cell Mol Life Sci. 2019 Sep;76(17):3349-3361. doi: 10.1007/s00018-019-03128-y. Epub 2019 May 9. Cell Mol Life Sci. 2019. PMID: 31073744 Free PMC article. Review.
Cited by
-
The small GTPase RhoB regulates TNFα signaling in endothelial cells.PLoS One. 2013 Sep 26;8(9):e75031. doi: 10.1371/journal.pone.0075031. eCollection 2013. PLoS One. 2013. PMID: 24086429 Free PMC article.
-
PLCβ3 mediates cortactin interaction with WAVE2 in MCP1-induced actin polymerization and cell migration.Mol Biol Cell. 2015 Dec 15;26(25):4589-606. doi: 10.1091/mbc.E15-08-0570. Epub 2015 Oct 21. Mol Biol Cell. 2015. PMID: 26490115 Free PMC article.
-
Autotaxin induces lung epithelial cell migration through lysoPLD activity-dependent and -independent pathways.Biochem J. 2011 Oct 1;439(1):45-55. doi: 10.1042/BJ20110274. Biochem J. 2011. PMID: 21696367 Free PMC article.
-
PI(3,5)P2 controls endosomal branched actin dynamics by regulating cortactin-actin interactions.J Cell Biol. 2015 Aug 31;210(5):753-69. doi: 10.1083/jcb.201412127. J Cell Biol. 2015. PMID: 26323691 Free PMC article.
-
Annexin A1 and A2: roles in retrograde trafficking of Shiga toxin.PLoS One. 2012;7(7):e40429. doi: 10.1371/journal.pone.0040429. Epub 2012 Jul 6. PLoS One. 2012. PMID: 22792315 Free PMC article.
References
-
- Aderem A. Signal transduction and the actin cytoskeleton: the roles of MARCKS and profilin. Trends Biochem. Sci. 1992;17:438–443. - PubMed
-
- Apodaca G. Endocytic traffic in polarized epithelial cells: role of the actin and microtubule cytoskeleton. Traffic. 2001;2:149–159. - PubMed
-
- Apodaca G., Enrich C., Mostov K. E. The calmodulin antagonist, W-13, alters transcytosis, recycling, and the morphology of the endocytic pathway in Madin-Darby canine kidney cells. J. Biol. Chem. 1994;269:19005–19013. - PubMed
-
- Ayscough K. R. Endocytosis: actin in the driving seat. Curr. Biol. 2004;14:R124–R126. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

