Viroporins from RNA viruses induce caspase-dependent apoptosis
- PMID: 17961183
- PMCID: PMC7162385
- DOI: 10.1111/j.1462-5822.2007.01057.x
Viroporins from RNA viruses induce caspase-dependent apoptosis
Abstract
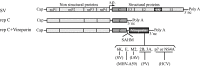
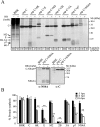
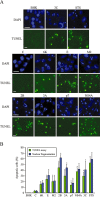
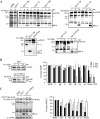
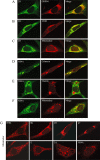
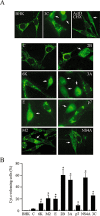
The virus-encoded viroporins are known to modify membrane permeability and play an essential role in virus budding. Here, a comparative analysis of the membrane permeabilization capacity of a number of viroporins was performed in baby hamster kidney cells. Synthesis of 6K protein from Sindbis virus, E from mouse hepatitis virus, M2 from influenza A virus, and 2B and 3A from poliovirus enhanced membrane permeability to different extents. We show that two proteins from hepatitis C virus, p7 and NS4A, also display viroporin activity to a level comparable to 6K protein. In addition to their capacity to disrupt ionic cellular homeostasis and promote bacterial cell lysis, the expressed viroporins were able to induce cell death. Degradation of internucleosomal DNA and generation of apoptotic bodies were observed upon viroporin expression. Consistently, cleavage of translation initiation factor 4GI and poly-(ADP-ribose) polymerase indicated activation of effector caspase-3. We found that poliovirus 2B localizes partially in mitochondria and induces an anomalous perinuclear distribution of these organelles. Mitochondria morphology was also altered after expression of other viroporins. Finally, detection of cytochrome c release from mitochondria suggests involvement of the mitochondrial pathway in viroporin-induced apoptosis. These findings suggest that viroporins induce caspase-dependent programmed cell death.
Figures
Similar articles
-
Cell permeabilization by poliovirus 2B viroporin triggers bystander permeabilization in neighbouring cells through a mechanism involving gap junctions.Cell Microbiol. 2010 Aug;12(8):1144-57. doi: 10.1111/j.1462-5822.2010.01460.x. Epub 2010 Mar 12. Cell Microbiol. 2010. PMID: 20331640 Free PMC article.
-
HCV non-structural NS4A protein of genotype 3a induces mitochondria mediated death by activating Bax and the caspase cascade.Microb Pathog. 2018 Nov;124:346-355. doi: 10.1016/j.micpath.2018.08.065. Epub 2018 Sep 1. Microb Pathog. 2018. PMID: 30179714
-
Synthetic 1,4-anthracenedione analogs induce cytochrome c release, caspase-9, -3, and -8 activities, poly(ADP-ribose) polymerase-1 cleavage and internucleosomal DNA fragmentation in HL-60 cells by a mechanism which involves caspase-2 activation but not Fas signaling.Biochem Pharmacol. 2004 Feb 1;67(3):523-37. doi: 10.1016/j.bcp.2003.09.012. Biochem Pharmacol. 2004. PMID: 15037204
-
Viroporins.FEBS Lett. 2003 Sep 18;552(1):28-34. doi: 10.1016/s0014-5793(03)00780-4. FEBS Lett. 2003. PMID: 12972148 Review.
-
Mechanisms of Apoptosis.Biochemistry (Mosc). 2015 Nov;80(11):1393-405. doi: 10.1134/S0006297915110012. Biochemistry (Mosc). 2015. PMID: 26615431 Review.
Cited by
-
Enterovirus 71 protease 2Apro targets MAVS to inhibit anti-viral type I interferon responses.PLoS Pathog. 2013 Mar;9(3):e1003231. doi: 10.1371/journal.ppat.1003231. Epub 2013 Mar 21. PLoS Pathog. 2013. PMID: 23555247 Free PMC article.
-
Structural and Functional Properties of the Hepatitis C Virus p7 Viroporin.Viruses. 2015 Aug 6;7(8):4461-81. doi: 10.3390/v7082826. Viruses. 2015. PMID: 26258788 Free PMC article. Review.
-
The FDA-Approved Drug Nelfinavir Inhibits Lytic Cell-Free but Not Cell-Associated Nonlytic Transmission of Human Adenovirus.Antimicrob Agents Chemother. 2020 Aug 20;64(9):e01002-20. doi: 10.1128/AAC.01002-20. Print 2020 Aug 20. Antimicrob Agents Chemother. 2020. PMID: 32601166 Free PMC article.
-
Antiapoptotic activity of the cardiovirus leader protein, a viral "security" protein.J Virol. 2009 Jul;83(14):7273-84. doi: 10.1128/JVI.00467-09. Epub 2009 May 6. J Virol. 2009. PMID: 19420082 Free PMC article.
-
The p7 protein of hepatitis C virus forms structurally plastic, minimalist ion channels.PLoS Comput Biol. 2012;8(9):e1002702. doi: 10.1371/journal.pcbi.1002702. Epub 2012 Sep 20. PLoS Comput Biol. 2012. PMID: 23028296 Free PMC article.
References
-
- Agirre, A. , Barco, A. , Carrasco, L. , and Nieva, J.L. (2002) Viroporin‐mediated membrane permeabilization. Pore formation by nonstructural poliovirus 2B protein. J Biol Chem 277: 40434–40441. - PubMed
-
- Agol, V.I. , Belov, G.A. , Bienz, K. , Egger, D. , Kolesnikova, M.S. , Raikhlin, N.T. , et al. (1998) Two types of death of poliovirus‐infected cells: caspase involvement in the apoptosis but not cytopathic effect. Virology 252: 343–353. - PubMed
-
- Aldabe, R. , Barco, A. , and Carrasco, L. (1996) Membrane permeabilization by poliovirus proteins 2B and 2BC. J Biol Chem 271: 23134–23137. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

