Sialic acid receptor detection in the human respiratory tract: evidence for widespread distribution of potential binding sites for human and avian influenza viruses
- PMID: 17961210
- PMCID: PMC2169242
- DOI: 10.1186/1465-9921-8-73
Sialic acid receptor detection in the human respiratory tract: evidence for widespread distribution of potential binding sites for human and avian influenza viruses
Abstract
Background: Influenza virus binds to cell receptors via sialic acid (SA) linked glycoproteins. They recognize SA on host cells through their haemagglutinins (H). The distribution of SA on cell surfaces is one determinant of host tropism and understanding its expression on human cells and tissues is important for understanding influenza pathogenesis. The objective of this study therefore was to optimize the detection of alpha2,3-linked and alpha2,6-linked SA by lectin histochemistry by investigating the binding of Sambucus nigra agglutinin (SNA) for SAalpha2,6Gal and Maackia amurensis agglutinin (MAA) for SAalpha2,3Gal in the respiratory tract of normal adults and children.
Methods: We used fluorescent and biotinylated SNA and MAA from different suppliers on archived and prospectively collected biopsy and autopsy specimens from the nasopharynx, trachea, bronchus and lungs of fetuses, infants and adults. We compared different methods of unmasking for tissue sections to determine if these would affect lectin binding. Using serial sections we then compared the lectin binding of MAA from different suppliers.
Results: We found that unmasking using microwave treatment in citrate buffer produced increased lectin binding to the ciliated and glandular epithelium of the respiratory tract. In addition we found that there were differences in tissue distribution of the alpha2,3 linked SA when 2 different isoforms of MAA (MAA1 and MAA2) lectin were used. MAA1 had widespread binding throughout the upper and lower respiratory tract and showed more binding to the respiratory epithelium of children than in adults. By comparison, MAA2 binding was mainly restricted to the alveolar epithelial cells of the lung with weak binding to goblet cells. SNA binding was detected in bronchial and alveolar epithelial cells and binding of this lectin was stronger to the paediatric epithelium compared to adult epithelium. Furthermore, the MAA lectins from 2 suppliers (Roche and EY Labs) tended to only bind in a pattern similar to MAA1 (Vector Labs) and produced a different binding pattern to MAA2 from Vector Labs.
Conclusion: The lectin binding pattern of MAA may vary depending on the supplier and the different isoforms of MAA show a different tissue distribution in the respiratory tract. This finding is important if conclusions about the potential binding sites of SAalpha2,3 binding viruses, such as influenza or human parainfluenza are to be made.
Figures



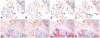
References
-
- Lamblin G, Lhermitte M, Klein A, Roussel P, Van Halbeek H, Vliegenthart JF. Biochem Soc Trans. 1984/08/01. 4. Vol. 12. 1984. Carbohydrate chains from human bronchial mucus glycoproteins: a wide spectrum of oligosaccharide structures; pp. 599–600. - PubMed
-
- Baum LG, Paulson JC. Acta Histochem Suppl. 1990/01/01. Vol. 40. 1990. Sialyloligosaccharides of the respiratory epithelium in the selection of human influenza virus receptor specificity; pp. 35–38. - PubMed
-
- Delmotte P, Degroote S, Merten MD, Van Seuningen I, Bernigaud A, Figarella C, Roussel P, Perini JM. Glycoconj J. 2002/06/27. 6. Vol. 18. 2001. Influence of TNFalpha on the sialylation of mucins produced by a transformed cell line MM-39 derived from human tracheal gland cells; pp. 487–497. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

