Absence of humoral mediated 5'AMP-activated protein kinase activation in human skeletal muscle and adipose tissue during exercise
- PMID: 17962330
- PMCID: PMC2375521
- DOI: 10.1113/jphysiol.2007.143123
Absence of humoral mediated 5'AMP-activated protein kinase activation in human skeletal muscle and adipose tissue during exercise
Abstract
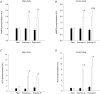
5'AMP-activated protein kinase (AMPK) exists as a heterotrimer comprising a catalytic alpha subunit and regulatory beta and gamma subunits. The AMPK system is activated under conditions of cellular stress, indicated by an increase in the AMP/ATP ratio, as observed, e.g. in muscles during contractile activity. AMPK was originally thought to be activated only by local intracellular mechanisms. However, recently it has become apparent that AMPK in mammals is also regulated by humoral substances, e.g. catecholamines. We studied whether humoral factors released during exercise regulate AMPK activity in contracting and resting muscles as well as in abdominal subcutaneous adipose tissue in humans. In resting leg muscle and adipose tissue the AMPK activity was not up-regulated by humoral factors during one-legged knee extensor exercise even when arm cranking exercise, inducing a approximately 20-fold increase in plasma catecholamine level, was added simultaneously. In exercising leg muscle the AMPK activity was increased by one-legged knee extensor exercise eliciting a whole body respiratory load of only 30% .VO(2,peak) but was not further increased by adding arm cranking exercise. In conclusion, during exercise with combined leg kicking and arm cranking, the AMPK activity in human skeletal muscle is restricted to contracting muscle without influence of marked increased catecholamine levels. Also, with this type of exercise the catecholamines or other humoral factors do not seem to be physiological regulators of AMPK in the subcutaneous adipose tissue.
Figures
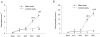
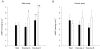

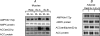
Similar articles
-
Regulation of 5'AMP-activated protein kinase activity and substrate utilization in exercising human skeletal muscle.Am J Physiol Endocrinol Metab. 2003 Apr;284(4):E813-22. doi: 10.1152/ajpendo.00436.2002. Epub 2002 Dec 17. Am J Physiol Endocrinol Metab. 2003. PMID: 12488245 Clinical Trial.
-
5'-AMP-activated protein kinase activity and subunit expression in exercise-trained human skeletal muscle.J Appl Physiol (1985). 2003 Feb;94(2):631-41. doi: 10.1152/japplphysiol.00642.2002. Epub 2002 Oct 11. J Appl Physiol (1985). 2003. PMID: 12391032
-
Role of 5'AMP-activated protein kinase in glycogen synthase activity and glucose utilization: insights from patients with McArdle's disease.J Physiol. 2002 Jun 15;541(Pt 3):979-89. doi: 10.1113/jphysiol.2002.018044. J Physiol. 2002. PMID: 12068056 Free PMC article.
-
Transgenic models--a scientific tool to understand exercise-induced metabolism: the regulatory role of AMPK (5'-AMP-activated protein kinase) in glucose transport and glycogen synthase activity in skeletal muscle.Biochem Soc Trans. 2003 Dec;31(Pt 6):1290-4. doi: 10.1042/bst0311290. Biochem Soc Trans. 2003. PMID: 14641045 Review.
-
AMP-activated protein kinase regulation and action in skeletal muscle during exercise.Biochem Soc Trans. 2003 Feb;31(Pt 1):191-5. doi: 10.1042/bst0310191. Biochem Soc Trans. 2003. PMID: 12546683 Review.
Cited by
-
Effects of High-Intensity Exercise Training on Adipose Tissue Mass, Glucose Uptake and Protein Content in Pre- and Post-menopausal Women.Front Sports Act Living. 2020 Jun 17;2:60. doi: 10.3389/fspor.2020.00060. eCollection 2020. Front Sports Act Living. 2020. PMID: 33345051 Free PMC article.
-
Fatty acid-inducible ANGPTL4 governs lipid metabolic response to exercise.Proc Natl Acad Sci U S A. 2014 Mar 18;111(11):E1043-52. doi: 10.1073/pnas.1400889111. Epub 2014 Mar 3. Proc Natl Acad Sci U S A. 2014. PMID: 24591600 Free PMC article.
-
The Regulation of Fat Metabolism During Aerobic Exercise.Biomolecules. 2020 Dec 21;10(12):1699. doi: 10.3390/biom10121699. Biomolecules. 2020. PMID: 33371437 Free PMC article. Review.
-
AMPK and the biochemistry of exercise: implications for human health and disease.Biochem J. 2009 Mar 1;418(2):261-75. doi: 10.1042/BJ20082055. Biochem J. 2009. PMID: 19196246 Free PMC article. Review.
-
Exercise and GLUT4 in human subcutaneous adipose tissue.Physiol Rep. 2018 Nov;6(22):e13918. doi: 10.14814/phy2.13918. Physiol Rep. 2018. PMID: 30450826 Free PMC article.
References
-
- Abu-Elheiga L, Almarza-Ortega DB, Baldini A, Wakil SJ. Human acetyl-CoA carboxylase 2. Molecular cloning, characterization, chromosomal mapping, and evidence for two isoforms. J Biol Chem. 1997;272:10669–10677. - PubMed
-
- Andersen P, Adams RP, Sjogaard G, Thorboe A, Saltin B. Dynamic knee extension as model for study of isolated exercising muscle in humans. J Appl Physiol. 1985;59:1647–1653. - PubMed
-
- Bandyopadhyay GK, Yu JG, Ofrecio J, Olefsky JM. Increased malonyl-CoA levels in muscle from obese and type 2 diabetic subjects lead to decreased fatty acid oxidation and increased lipogenesis; thiazolidinedione treatment reverses these defects. Diabetes. 2006;55:2277–2285. - PubMed
-
- Beauloye C, Marsin AS, Bertrand L, Krause U, Hardie DG, Vanoverschelde JL, Hue L. Insulin antagonizes AMP-activated protein kinase activation by ischemia or anoxia in rat hearts, without affecting total adenine nucleotides. FEBS Lett. 2001;505:348–352. - PubMed
-
- Carling D, Clarke PR, Zammit VA, Hardie DG. Purification and characterization of the AMP-activated protein kinase. Copurification of acetyl-CoA carboxylase kinase and 3-hydroxy-3-methylglutaryl-CoA reductase kinase activities. Eur J Biochem. 1989;186:129–136. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

