Reactivity and biological activity of the marine sesquiterpene hydroquinone avarol and related compounds from sponges of the order Dictyoceratida
- PMID: 17962742
- PMCID: PMC6148525
- DOI: 10.3390/11010001
Reactivity and biological activity of the marine sesquiterpene hydroquinone avarol and related compounds from sponges of the order Dictyoceratida
Abstract
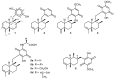
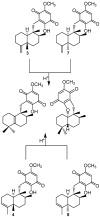
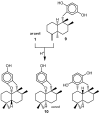
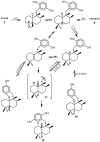
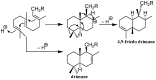
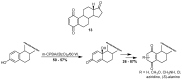
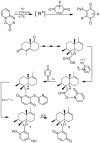
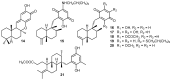
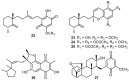
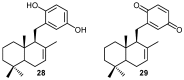
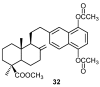
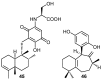
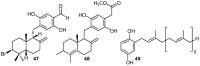
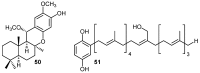
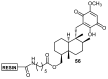
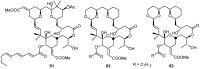
A review of results of bioactivity and reactivity examinations of marine sesquiterpene (hydro)quinones is presented. The article is focused mostly on friedo- rearranged drimane structural types, isolated from sponges of the order Dictyoceratida. Examples of structural correlations are outlined. Available results on the mechanism of redox processes and examinations of chemo- and regioselectivity in addition reactions are presented and, where possible, analyzed in relation to established bioactivities. Most of the bioactivity examinations are concerned with antitumor activities and the mechanism thereof, such as DNA damage, arylation of nucleophiles, tubulin assembly inhibition, protein kinase inhibition, inhibition of the arachidonic cascade, etc. Perspectives on marine drug development are discussed with respect to biotechnological methods and synthesis. Examples of the recognition of validated core structures and synthesis of structurally simplified compounds retaining modes of activity are analyzed.
Figures
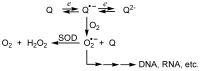
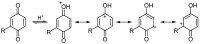
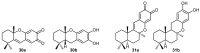
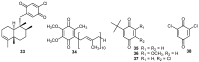
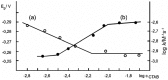
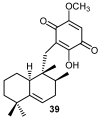
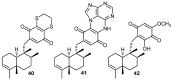
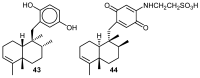
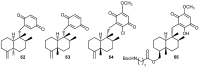
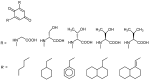
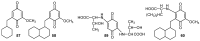
Similar articles
-
Cytotoxic terpene quinones from marine sponges.Mar Drugs. 2010 Nov 24;8(12):2849-70. doi: 10.3390/md8122849. Mar Drugs. 2010. PMID: 21339953 Free PMC article. Review.
-
New sesquiterpene hydroquinones from a Taiwanese marine sponge, Hippospongia metachromia.J Nat Prod. 2001 Jun;64(6):801-3. doi: 10.1021/np000610c. J Nat Prod. 2001. PMID: 11421749
-
Pleiotropic Role of Puupehenones in Biomedical Research.Mar Drugs. 2017 Oct 21;15(10):325. doi: 10.3390/md15100325. Mar Drugs. 2017. PMID: 29065486 Free PMC article. Review.
-
Sesquiterpene Quinones/Hydroquinones from the Marine Sponge Spongia pertusa Esper.J Nat Prod. 2017 May 26;80(5):1436-1445. doi: 10.1021/acs.jnatprod.6b01105. Epub 2017 Apr 11. J Nat Prod. 2017. PMID: 28398051
-
Application of cell culture for the production of bioactive compounds from sponges: synthesis of avarol by primmorphs from Dysidea avara.J Nat Prod. 2000 Aug;63(8):1077-81. doi: 10.1021/np000003p. J Nat Prod. 2000. PMID: 10978201
Cited by
-
Oxidative radical coupling of hydroquinones and thiols using chromic acid: one-pot synthesis of quinonyl alkyl/aryl thioethers.RSC Adv. 2020 May 21;10(33):19454-19462. doi: 10.1039/d0ra01519a. eCollection 2020 May 20. RSC Adv. 2020. PMID: 35515459 Free PMC article.
-
Plant growth promoting endophyte Burkholderia contaminans NZ antagonizes phytopathogen Macrophomina phaseolina through melanin synthesis and pyrrolnitrin inhibition.PLoS One. 2021 Sep 30;16(9):e0257863. doi: 10.1371/journal.pone.0257863. eCollection 2021. PLoS One. 2021. PMID: 34591915 Free PMC article.
-
Synthesis, Anticancer Activity, and Docking Studies of Novel Hydroquinone-Chalcone-Pyrazoline Hybrid Derivatives.Int J Mol Sci. 2024 Jul 2;25(13):7281. doi: 10.3390/ijms25137281. Int J Mol Sci. 2024. PMID: 39000394 Free PMC article.
-
Comparison of the biological properties of several marine sponge-derived sesquiterpenoid quinones.Molecules. 2007 Jul 11;12(7):1376-88. doi: 10.3390/12071376. Molecules. 2007. PMID: 17909493 Free PMC article.
-
Cyclodepsipeptides from marine sponges: natural agents for drug research.Mar Drugs. 2010 Mar 22;8(3):810-34. doi: 10.3390/md8030810. Mar Drugs. 2010. PMID: 20411126 Free PMC article.
References
-
- Berquist P.R. Sponges. Hutchinson; London: 1978.
-
- Simpson T.L. The cell biology of sponges. Springer; New York: 1984.
-
- Faulkner D.J. Marine natural products. Nat. Prod. Rep. 2002;19:1–48. - PubMed
-
and earlier reviews in this series.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

