U2 snRNP binds intronless histone pre-mRNAs to facilitate U7-snRNP-dependent 3' end formation
- PMID: 17964263
- PMCID: PMC2149891
- DOI: 10.1016/j.molcel.2007.09.026
U2 snRNP binds intronless histone pre-mRNAs to facilitate U7-snRNP-dependent 3' end formation
Abstract
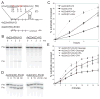
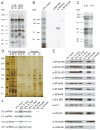
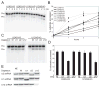
In metazoa, pre-mRNA 3' end formation occurs via two pathways: cleavage/polyadenylation for the majority of RNA polymerase II transcripts and U7-snRNP-dependent cleavage for replication-dependent histone pre-mRNAs. An RNA element derived from a replication-dependent histone gene affects multiple steps of pre-mRNA processing. Here, we demonstrate that a portion of this RNA element, present in the majority of histone mRNAs, stimulates U7-snRNP-dependent cleavage. Surprisingly, this element binds U2 snRNP, although it is derived from an intronless mRNA. Specifically, SF3b, a U2 and U12-snRNP component, contacts the RNA element both in vitro and in vivo in conjunction with hPrp43, a DEAH-box helicase. Tethering either U2 or U12 snRNP to histone pre-mRNA substrates stimulates U7-snRNP-dependent cleavage in vitro and in vivo. Finally, we show that U2 snRNP associates with histone pre-mRNAs in vivo. We conclude that U2 snRNP plays a nonsplicing role in histone mRNA maturation.
Figures



Comment in
-
U2 snRNP: not just for poly(A) mRNAs.Mol Cell. 2007 Nov 9;28(3):353-4. doi: 10.1016/j.molcel.2007.10.015. Mol Cell. 2007. PMID: 17996698 Review.
Similar articles
-
U2 snRNP: not just for poly(A) mRNAs.Mol Cell. 2007 Nov 9;28(3):353-4. doi: 10.1016/j.molcel.2007.10.015. Mol Cell. 2007. PMID: 17996698 Review.
-
3'-End processing of histone pre-mRNAs in Drosophila: U7 snRNP is associated with FLASH and polyadenylation factors.RNA. 2013 Dec;19(12):1726-44. doi: 10.1261/rna.040360.113. Epub 2013 Oct 21. RNA. 2013. PMID: 24145821 Free PMC article.
-
The 68 kDa subunit of mammalian cleavage factor I interacts with the U7 small nuclear ribonucleoprotein and participates in 3'-end processing of animal histone mRNAs.Nucleic Acids Res. 2010 Nov;38(21):7637-50. doi: 10.1093/nar/gkq613. Epub 2010 Jul 15. Nucleic Acids Res. 2010. PMID: 20634199 Free PMC article.
-
U7 snRNP is recruited to histone pre-mRNA in a FLASH-dependent manner by two separate regions of the stem-loop binding protein.RNA. 2017 Jun;23(6):938-951. doi: 10.1261/rna.060806.117. Epub 2017 Mar 13. RNA. 2017. PMID: 28289156 Free PMC article.
-
Formation of the 3' end of histone mRNA.Gene. 1999 Oct 18;239(1):1-14. doi: 10.1016/s0378-1119(99)00367-4. Gene. 1999. PMID: 10571029 Review.
Cited by
-
Application of a novel strategy of engineering conditional alleles to a single exon gene, Sox2.PLoS One. 2012;7(9):e45768. doi: 10.1371/journal.pone.0045768. Epub 2012 Sep 27. PLoS One. 2012. PMID: 23029233 Free PMC article.
-
Cajal body function in genome organization and transcriptome diversity.Bioessays. 2016 Dec;38(12):1197-1208. doi: 10.1002/bies.201600144. Epub 2016 Oct 21. Bioessays. 2016. PMID: 27767214 Free PMC article. Review.
-
Termination of pre-mRNA splicing requires that the ATPase and RNA unwindase Prp43p acts on the catalytic snRNA U6.Genes Dev. 2019 Nov 1;33(21-22):1555-1574. doi: 10.1101/gad.328294.119. Epub 2019 Sep 26. Genes Dev. 2019. PMID: 31558568 Free PMC article.
-
A core complex of CPSF73, CPSF100, and Symplekin may form two different cleavage factors for processing of poly(A) and histone mRNAs.Mol Cell. 2009 May 15;34(3):322-32. doi: 10.1016/j.molcel.2009.04.024. Mol Cell. 2009. PMID: 19450530 Free PMC article.
-
Noncoding RNA-chromatin association: Functions and mechanisms.Fundam Res. 2023 Apr 7;3(5):665-675. doi: 10.1016/j.fmre.2023.03.006. eCollection 2023 Sep. Fundam Res. 2023. PMID: 38933302 Free PMC article. Review.
References
-
- Barnard DC, Ryan K, Manley JL, Richter JD. Symplekin and xGLD-2 are required for CPEB-mediated cytoplasmic polyadenylation. Cell. 2004;119:641–651. - PubMed
-
- Blanchette M, Labourier E, Green RE, Brenner SE, Rio DC. Genome-wide analysis reveals an unexpected function for the Drosophila splicing factor U2AF50 in the nuclear export of intronless mRNAs. Mol Cell. 2004;14:775–786. - PubMed
-
- Bond UM, Yario TA, Steitz JA. Multiple processing-defective mutations in a mammalian histone pre-mRNA are suppressed by compensatory changes in U7 RNA both in vivo and in vitro. Genes Dev. 1991;5:1709–1722. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

