A novel method for generating and screening peptides and libraries displayed on adenovirus fiber
- PMID: 17965092
- PMCID: PMC2175307
- DOI: 10.1093/nar/gkm914
A novel method for generating and screening peptides and libraries displayed on adenovirus fiber
Abstract
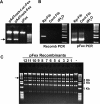
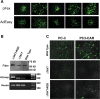
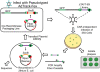
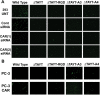
Capsid-displayed adenoviral peptide libraries have been a significant, yet unfeasible goal in biotechnology. Three barriers have made this difficult: the large size of the viral genome, the low efficiency of converting plasmid-based genomes into packaged adenovirus and the fact that library amplification is hampered by the ability of two (or more) virus to co-infect one cell. Here, we present a novel vector system, pFex, which is capable of overcoming all three barriers. With pFex, modified fiber genes are recombined into the natural genetic locus of adenovirus through unidirectional Cre-lox recombination. Modified-fiber genes can be directly shuttled into replicating viral genomes in mammalian cells. The 'acceptor' vector does not contain the fiber gene, and therefore does not propagate until it has received a 'donor' fiber gene. Therefore, This methodology overcomes the low efficiency of transfecting large viral genomes and bypasses the need for transition to functional virus. Thus, with a fiber-shuttle library, one can generate and evaluate large numbers of fiber-modified adenovirus simultaneously. Finally, successful fiber genes can be rescued from virus and recombined back into shuttle plasmids, avoiding the need to propagate mixed viral pools. For proof of principal, we use this new system to screen a capsid-displayed peptide library for retargeted viral infection.
Figures


References
-
- Wickham TJ, Roelvink PW, Brough DE, Kovesdi I. Adenovirus targeted to heparan-containing receptors increases its gene delivery efficiency to multiple cell types. Nat. Biotechnol. 1996;14:1570–1573. - PubMed
-
- Wickham TJ, Carrion ME, Kovesdi I. Targeting of adenovirus penton base to new receptors through replacement of its RGD motif with other receptor-specific peptide motifs. Gene Ther. 1995;2:750–756. - PubMed
-
- Kurachi S, Koizumi N, Sakurai F, Kawabata K, Sakurai H, Nakagawa S, Hayakawa T, Mizuguchi H. Characterization of capsid-modified adenovirus vectors containing heterologous peptides in the fiber knob, protein IX, or hexon. Gene Ther. 2006 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

