Intermedin (adrenomedullin-2): a novel counter-regulatory peptide in the cardiovascular and renal systems
- PMID: 17965749
- PMCID: PMC2268039
- DOI: 10.1038/sj.bjp.0707494
Intermedin (adrenomedullin-2): a novel counter-regulatory peptide in the cardiovascular and renal systems
Abstract
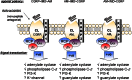
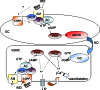
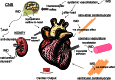
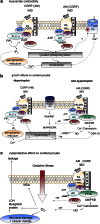
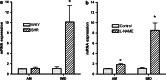
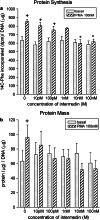
Intermedin (IMD) is a novel peptide related to calcitonin gene-related peptide (CGRP) and adrenomedullin (AM). Proteolytic processing of a larger precursor yields a series of biologically active C-terminal fragments, IMD(1-53), IMD(1-47) and IMD(8-47). IMD shares a family of receptors with AM and CGRP composed of a calcitonin-receptor like receptor (CALCRL) associated with one of three receptor activity modifying proteins (RAMP). Compared to CGRP, IMD is less potent at CGRP(1) receptors but more potent at AM(1) receptors and AM(2) receptors; compared to AM, IMD is more potent at CGRP(1) receptors but less potent at AM(1) and AM(2) receptors. The cellular and tissue distribution of IMD overlaps in some aspects with that of CGRP and AM but is distinct from both. IMD is present in neonatal but absent or expressed sparsely, in adult heart and vasculature and present at low levels in plasma. The prominent localization of IMD in hypothalamus and pituitary and in kidney is consistent with a physiological role in the central and peripheral regulation of the circulation and water-electrolyte homeostasis. IMD is a potent systemic and pulmonary vasodilator, influences regional blood flow and augments cardiac contractility. IMD protects myocardium from the deleterious effects of oxidative stress associated with ischaemia-reperfusion injury and exerts an anti-growth effect directly on cardiomyocytes to oppose the influence of hypertrophic stimuli. The robust increase in expression of the peptide in hypertrophied and ischaemic myocardium indicates an important protective role for IMD as an endogenous counter-regulatory peptide in the heart.
Figures

References
-
- Abdelrahman AM, Pang CCY. Effect of intermedin/adrenomedullin-2 on venous tone in conscious rats. Naunyn Schmied Arch Pharmacol. 2006;373:376–380. - PubMed
-
- Alevizaki M, Shiraishi A, Rassool FV, Ferrier GJM, Macintyre I, Legon S. The calcitonin-like sequence if the β-CGRP gene. FEBS Lett. 1986;206:47–52. - PubMed
-
- Autelitano DJ, Ridings R. Adrenomedullin signalling in cardiomyocytes is dependent upon CRLR and RAMP2 expression. Peptides. 2001;22:1851–1857. - PubMed
-
- Bell D, McDermott BJ. CGRP stimulates a positive contractile response in rat ventricular cardiomyocytes. J Cardiovasc Pharmacol. 1994;23:1011–1021. - PubMed
-
- Bell D, McDermott BJ. Calcitonin gene-related peptide in the cardiovascular system: characterisation of receptor populations and their pathophysiological significance. Pharmacol Rev. 1996;48:253–288. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

