Cerebellar motor learning: when is cortical plasticity not enough?
- PMID: 17967048
- PMCID: PMC2041974
- DOI: 10.1371/journal.pcbi.0030197
Cerebellar motor learning: when is cortical plasticity not enough?
Abstract
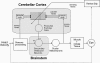
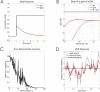
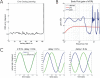
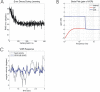
Classical Marr-Albus theories of cerebellar learning employ only cortical sites of plasticity. However, tests of these theories using adaptive calibration of the vestibulo-ocular reflex (VOR) have indicated plasticity in both cerebellar cortex and the brainstem. To resolve this long-standing conflict, we attempted to identify the computational role of the brainstem site, by using an adaptive filter version of the cerebellar microcircuit to model VOR calibration for changes in the oculomotor plant. With only cortical plasticity, introducing a realistic delay in the retinal-slip error signal of 100 ms prevented learning at frequencies higher than 2.5 Hz, although the VOR itself is accurate up to at least 25 Hz. However, the introduction of an additional brainstem site of plasticity, driven by the correlation between cerebellar and vestibular inputs, overcame the 2.5 Hz limitation and allowed learning of accurate high-frequency gains. This "cortex-first" learning mechanism is consistent with a wide variety of evidence concerning the role of the flocculus in VOR calibration, and complements rather than replaces the previously proposed "brainstem-first" mechanism that operates when ocular tracking mechanisms are effective. These results (i) describe a process whereby information originally learnt in one area of the brain (cerebellar cortex) can be transferred and expressed in another (brainstem), and (ii) indicate for the first time why a brainstem site of plasticity is actually required by Marr-Albus type models when high-frequency gains must be learned in the presence of error delay.
Conflict of interest statement
Figures

Similar articles
-
Chronic changes in inputs to dorsal Y neurons accompany VOR motor learning.J Neurophysiol. 2006 Mar;95(3):1812-25. doi: 10.1152/jn.01061.2005. Epub 2005 Nov 30. J Neurophysiol. 2006. PMID: 16319196
-
Report on a workshop concerning the cerebellum and motor learning, held in St Louis October 2004.Cerebellum. 2005;4(2):140-50. doi: 10.1080/14734220510007987. Cerebellum. 2005. PMID: 16035196
-
Computational Theory Underlying Acute Vestibulo-ocular Reflex Motor Learning with Cerebellar Long-Term Depression and Long-Term Potentiation.Cerebellum. 2017 Aug;16(4):827-839. doi: 10.1007/s12311-017-0857-6. Cerebellum. 2017. PMID: 28444617
-
Learning in a simple motor system.Learn Mem. 2004 Mar-Apr;11(2):127-36. doi: 10.1101/lm.65804. Learn Mem. 2004. PMID: 15054127 Review.
-
Cerebellum-dependent learning: the role of multiple plasticity mechanisms.Annu Rev Neurosci. 2004;27:581-609. doi: 10.1146/annurev.neuro.27.070203.144238. Annu Rev Neurosci. 2004. PMID: 15217344 Review.
Cited by
-
Cerebellar inactivation impairs memory of learned prism gaze-reach calibrations.J Neurophysiol. 2011 May;105(5):2248-59. doi: 10.1152/jn.01009.2010. Epub 2011 Mar 9. J Neurophysiol. 2011. PMID: 21389311 Free PMC article.
-
Modeling memory consolidation during posttraining periods in cerebellovestibular learning.Proc Natl Acad Sci U S A. 2015 Mar 17;112(11):3541-6. doi: 10.1073/pnas.1413798112. Epub 2015 Mar 3. Proc Natl Acad Sci U S A. 2015. PMID: 25737547 Free PMC article.
-
Adaptive-filter models of the cerebellum: computational analysis.Cerebellum. 2008;7(4):567-71. doi: 10.1007/s12311-008-0067-3. Cerebellum. 2008. PMID: 18972182
-
A normative principle governing memory transfer in cerebellar motor learning.Nat Commun. 2025 Jul 1;16(1):5479. doi: 10.1038/s41467-025-60511-z. Nat Commun. 2025. PMID: 40595477 Free PMC article.
-
Optimal properties of analog perceptrons with excitatory weights.PLoS Comput Biol. 2013;9(2):e1002919. doi: 10.1371/journal.pcbi.1002919. Epub 2013 Feb 21. PLoS Comput Biol. 2013. PMID: 23436991 Free PMC article.
References
-
- Ito M. Neurophysiological aspects of the cerebellar motor control system. Intl J Neurol. 1970;7:162–176. - PubMed
-
- Ito M. The cerebellum and neural control. New York: Raven Press; 1984.
-
- Ito M. Cerebellar microcomplexes. Intl Rev Neurobiol. 1997;41:475–487. - PubMed
-
- Boyden ES, Katoh A, Raymond JL. Cerebellum-dependent learning: the role of multiple plasticity mechanisms. Annu Rev Neurosci. 2004;27:581–609. - PubMed
-
- Albus JS. A theory of cerebellar function. Math Biosci. 1971;10:25–61.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

