Identification of internal ribosome entry segment (IRES)-trans-acting factors for the Myc family of IRESs
- PMID: 17967896
- PMCID: PMC2223313
- DOI: 10.1128/MCB.01298-07
Identification of internal ribosome entry segment (IRES)-trans-acting factors for the Myc family of IRESs
Abstract
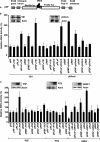
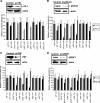
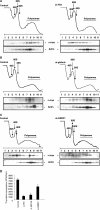
The proto-oncogenes c-, L-, and N-myc can all be translated by the alternative method of internal ribosome entry whereby the ribosome is recruited to a complex structural element (an internal ribosome entry segment [IRES]). Ribosome recruitment is dependent upon the presence of IRES-trans-acting factors (ITAFs) that act as RNA chaperones and allow the mRNA to attain the correct conformation for the interaction of the 40S subunit. One of the major challenges for researchers in this area is to determine whether there are groups of ITAFs that regulate the IRES-mediated translation of subsets of mRNAs. We have identified four proteins, termed GRSF-1 (G-rich RNA sequence binding factor 1), YB-1 (Y-box binding protein 1), PSF (polypyrimidine tract binding protein-associated splicing factor), and its binding partner, p54nrb, that bind to the myc family of IRESs. We show that these proteins positively regulate the translation of the Myc family of oncoproteins (c-, L-, and N-Myc) in vivo and in vitro. Interestingly, synthesis from the unrelated IRESs, BAG-1 and Apaf-1, was not affected by YB-1, GRSF-1, or PSF levels in vivo, suggesting that these three ITAFs are specific to the myc IRESs. Myc proteins play a role in cell proliferation; therefore, these results have important implications regarding the control of tumorigenesis.
Figures


References
-
- Aalto, Y., W. El-Rifai, L. Vilpo, J. Ollila, B. Nagy, M. Vihinen, J. Vilpo, and S. Knuutila. 2001. Distinct gene expression profiling in chronic lymphocytic leukemia with 11q23 deletion. Leukemia 151721-1728. - PubMed
-
- Bladen, C. L. 2005. Identification of the polypyrimidine tract binding protein-associated splicing factor·p54(nrb) complex as a candidate DNA double-strand break rejoining factor. J. Biol. Chem. 2805205-5210. - PubMed
-
- Bonnal, S., C. Schaeffer, L. Creancier, S. Clamens, H. Moine, A. C. Prats, and S. Vagner. 2003. A single internal ribosome entry site containing a G quartet RNA structure drives fibroblast growth factor 2 gene expression at four alternative translation initiation codons. J. Biol. Chem. 27839330-39336. - PMC - PubMed
-
- Brown, S. A., J. Ripperger, S. Kadener, F. Fleury-Olela, F. Vilbois, M. Rosbash, and U. Schibler. 2005. PERIOD1-associated proteins modulate the negative limb of the mammalian circadian oscillator. Science 308693-696. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
