Drosophila bestrophin-1 chloride current is dually regulated by calcium and cell volume
- PMID: 17968025
- PMCID: PMC2151665
- DOI: 10.1085/jgp.200709795
Drosophila bestrophin-1 chloride current is dually regulated by calcium and cell volume
Abstract
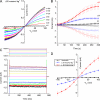
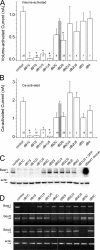
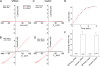
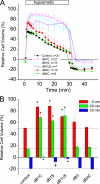
Mutations in the human bestrophin-1 (hBest1) gene are responsible for Best vitelliform macular dystrophy, however the mechanisms leading to retinal degeneration have not yet been determined because the function of the bestrophin protein is not fully understood. Bestrophins have been proposed to comprise a new family of Cl(-) channels that are activated by Ca(2+). While the regulation of bestrophin currents has focused on intracellular Ca(2+), little is known about other pathways/mechanisms that may also regulate bestrophin currents. Here we show that Cl(-) currents in Drosophila S2 cells, that we have previously shown are mediated by bestrophins, are dually regulated by Ca(2+) and cell volume. The bestrophin Cl(-) currents were activated in a dose-dependent manner by osmotic pressure differences between the internal and external solutions. The increase in the current was accompanied by cell swelling. The volume-regulated Cl(-) current was abolished by treating cells with each of four different RNAi constructs that reduced dBest1 expression. The volume-regulated current was rescued by transfecting with dBest1. Furthermore, cells not expressing dBest1 were severely depressed in their ability to regulate their cell volume. Volume regulation and Ca(2+) regulation can occur independently of one another: the volume-regulated current was activated in the complete absence of Ca(2+) and the Ca(2+)-activated current was activated independently of alterations in cell volume. These two pathways of bestrophin channel activation can interact; intracellular Ca(2+) potentiates the magnitude of the current activated by changes in cell volume. We conclude that in addition to being regulated by intracellular Ca(2+), Drosophila bestrophins are also novel members of the volume-regulated anion channel (VRAC) family that are necessary for cell volume homeostasis.
Figures

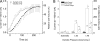

Similar articles
-
Single Cl- channels activated by Ca2+ in Drosophila S2 cells are mediated by bestrophins.J Gen Physiol. 2006 Sep;128(3):247-59. doi: 10.1085/jgp.200609581. J Gen Physiol. 2006. PMID: 16940553 Free PMC article.
-
Rescue of volume-regulated anion current by bestrophin mutants with altered charge selectivity.J Gen Physiol. 2008 Nov;132(5):537-46. doi: 10.1085/jgp.200810065. J Gen Physiol. 2008. PMID: 18955594 Free PMC article.
-
Drosophila bestrophin-1 currents are regulated by phosphorylation via a CaMKII dependent mechanism.PLoS One. 2013;8(3):e58875. doi: 10.1371/journal.pone.0058875. Epub 2013 Mar 12. PLoS One. 2013. PMID: 23554946 Free PMC article.
-
Molecular physiology of bestrophins: multifunctional membrane proteins linked to best disease and other retinopathies.Physiol Rev. 2008 Apr;88(2):639-72. doi: 10.1152/physrev.00022.2007. Physiol Rev. 2008. PMID: 18391176 Review.
-
Mechanisms of cellular synchronization in the vascular wall. Mechanisms of vasomotion.Dan Med Bull. 2010 Oct;57(10):B4191. Dan Med Bull. 2010. PMID: 21040688 Review.
Cited by
-
LRRC8A is essential for volume-regulated anion channel in smooth muscle cells contributing to cerebrovascular remodeling during hypertension.Cell Prolif. 2021 Dec;54(12):e13146. doi: 10.1111/cpr.13146. Epub 2021 Nov 1. Cell Prolif. 2021. PMID: 34725866 Free PMC article.
-
Chloride channels: often enigmatic, rarely predictable.Annu Rev Physiol. 2010;72:95-121. doi: 10.1146/annurev-physiol-021909-135811. Annu Rev Physiol. 2010. PMID: 19827947 Free PMC article. Review.
-
Bestrophin-encoded Ca²⁺-activated Cl⁻ channels underlie a current with properties similar to the native current in the moth Spodoptera littoralis olfactory receptor neurons.PLoS One. 2012;7(12):e52691. doi: 10.1371/journal.pone.0052691. Epub 2012 Dec 26. PLoS One. 2012. PMID: 23300744 Free PMC article.
-
SWELL1, a plasma membrane protein, is an essential component of volume-regulated anion channel.Cell. 2014 Apr 10;157(2):447-458. doi: 10.1016/j.cell.2014.03.024. Cell. 2014. PMID: 24725410 Free PMC article.
-
Calcium activates a chloride conductance likely involved in olfactory receptor neuron repolarization in the moth Spodoptera littoralis.J Neurosci. 2010 May 5;30(18):6323-33. doi: 10.1523/JNEUROSCI.0261-10.2010. J Neurosci. 2010. PMID: 20445058 Free PMC article.
References
-
- Adorante, J.S. 1995. Regulatory volume decrease in frog retinal pigment epithelium. Am. J. Physiol. 268:C89–C100. - PubMed
-
- Allikmets, R., J.M. Seddon, P.S. Bernstein, A. Hutchinson, A. Atkinson, S. Sharma, B. Gerrard, W. Li, M.L. Metzker, C. Wadelius, et al. 1999. Evaluation of the Best disease gene in patients with age-related macular degeneration and other maculopathies. Hum. Genet. 104:449–453. - PubMed
-
- Chen, B., G. Nicol, and W.K. Cho. 2007. Role of calcium in volume-activated chloride currents in a mouse cholangiocyte cell line. J. Membr. Biol. 215:1–13. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

