Prolonged-release nicotinic acid for the management of dyslipidemia: an update including results from the NAUTILUS study
- PMID: 17969377
- PMCID: PMC2291331
- DOI: 10.2147/vhrm.s1023
Prolonged-release nicotinic acid for the management of dyslipidemia: an update including results from the NAUTILUS study
Abstract
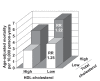
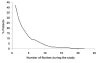
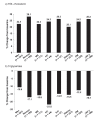
Low HDL-cholesterol (<1.02 mmol/L [40 mg/dL] in men or <1.29 mmol/L [50 mg/dL] in women) occurs in about one-third of European patients with dyslipidemia and is an independent cardiovascular risk factor. Simultaneous correction of low HDL-cholesterol and high total-cholesterol and LDL-cholesterol may provide reductions in cardiovascular morbidity and mortality beyond those possible with statins alone. Nicotinic acid (niacin in the US) is the most effective means of increasing HDL-cholesterol available and has been shown to reduce cardiovascular event rates significantly. Niaspan (prolonged-release nicotinic acid) provides a convenient, once-daily means of administering nicotinic acid. Clinical studies with Niaspan have demonstrated marked, long-term increases in HDL-cholesterol with additional useful benefits on triglycerides, LDL-cholesterol, and lipid sub-profiles. The NAUTILUS study demonstrated the beneficial efficacy and tolerability profiles of Niaspan in a usual-care setting. The most common side-effect of Niaspan is flushing, which infrequently causes treatment discontinuation and which usually subsides over continued treatment. The ARBITER 2 and ARBITER 3 studies showed 1-2 years of treatment with Niaspan plus a statin induced regression of atherosclerosis in patients with coronary artery disease. The effect of Niaspan-statin treatment, relative to a statin alone, on clinical cardiovascular outcomes is currently under evaluation. Niaspan represents a practical means of correcting low HDL-cholesterol, an independent risk factor for adverse cardiovascular outcomes.
Figures




Similar articles
-
Correction of low HDL cholesterol to reduce cardiovascular risk: practical considerations relating to the therapeutic use of prolonged-release nicotinic acid (Niaspan).Int J Clin Pract. 2007 Nov;61(11):1914-21. doi: 10.1111/j.1742-1241.2007.01514.x. Int J Clin Pract. 2007. PMID: 17935550 Review.
-
Efficacy and safety of fenofibric acid co-administered with low- or moderate-dose statin in patients with mixed dyslipidemia and type 2 diabetes mellitus: results of a pooled subgroup analysis from three randomized, controlled, double-blind trials.Am J Cardiovasc Drugs. 2010;10(2):73-84. doi: 10.2165/10061630-000000000-00000. Am J Cardiovasc Drugs. 2010. PMID: 20136164
-
Long-term safety and efficacy of a combination of niacin extended release and simvastatin in patients with dyslipidemia: the OCEANS study.Am J Cardiovasc Drugs. 2008;8(2):69-81. doi: 10.2165/00129784-200808020-00001. Am J Cardiovasc Drugs. 2008. PMID: 18422390 Clinical Trial.
-
Comparison of once-daily, niacin extended-release/lovastatin with standard doses of atorvastatin and simvastatin (the ADvicor Versus Other Cholesterol-Modulating Agents Trial Evaluation [ADVOCATE]).Am J Cardiol. 2003 Mar 15;91(6):667-72. doi: 10.1016/s0002-9149(03)00007-9. Am J Cardiol. 2003. PMID: 12633795 Clinical Trial.
-
Managing dyslipidemia in the high-risk patient.Am J Cardiol. 2002 Mar 7;89(5A):50C-57C. doi: 10.1016/s0002-9149(02)02229-4. Am J Cardiol. 2002. PMID: 11900720 Review.
Cited by
-
Hydroxybenzoic acid isomers and the cardiovascular system.Nutr J. 2014 Jun 19;13:63. doi: 10.1186/1475-2891-13-63. Nutr J. 2014. PMID: 24943896 Free PMC article. Review.
-
Partitioning of homologous nicotinic acid ester prodrugs (nicotinates) into dipalmitoylphosphatidylcholine (DPPC) membrane bilayers.Colloids Surf B Biointerfaces. 2010 Jun 15;78(1):75-84. doi: 10.1016/j.colsurfb.2010.02.015. Epub 2010 Feb 18. Colloids Surf B Biointerfaces. 2010. PMID: 20227859 Free PMC article.
-
Empowering Medical Students: Unveiling the Benefits of Nutrition Seminars and Plant-Based Diets in Medical School Education.Am J Lifestyle Med. 2024 Apr 3:15598276241242732. doi: 10.1177/15598276241242732. Online ahead of print. Am J Lifestyle Med. 2024. PMID: 39554927 Free PMC article.
-
Quinoa Seed Lowers Serum Triglycerides in Overweight and Obese Subjects: A Dose-Response Randomized Controlled Clinical Trial.Curr Dev Nutr. 2017 Aug 24;1(9):e001321. doi: 10.3945/cdn.117.001321. eCollection 2017 Sep. Curr Dev Nutr. 2017. PMID: 29955719 Free PMC article.
References
-
- AIM-HIGH trial, www.clinicaltrials.gov identifier NCT00120289
-
- Al-Sheikh AA, Karas RH. Relative safety of lipid altering drugs in the general population. Abstract 3707, presented at the 2005. American Heart Association Scientific Sessions; Nov 13–6, 2005; Dallas, Texas, USA. 2005.
-
- Assmann G, Schulte H, von Eckardstein A, et al. High-density lipoprotein cholesterol as a predictor of coronary heart disease risk. The PROCAM experience and pathophysiological implications for reverse cholesterol transport. Atherosclerosis. 1996;124(Supp l):S11–20. - PubMed
-
- Balkau B, Charles MA, Drivsholm T, et al. Frequency of the WHO metabolic syndrome in European cohorts, and an alternative definition of an insulin resistance syndrome. Diabetes Metab. 2002;28:364–76. - PubMed
-
- Bays HE, Dujovne CA, McGovern ME, et al. Comparison of once-daily, niacin extended-release/lovastatin with standard doses of atorvastatin and simvastatin (the ADvicor Versus Other Cholesterol-Modulating Agents Trial Evaluation [ADVOCATE]) Am J Cardiol. 2003;91:667–72. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

