Visual impairment and circadian rhythm disorders
- PMID: 17969867
- PMCID: PMC3202494
- DOI: 10.31887/DCNS.2007.9.3/slockley
Visual impairment and circadian rhythm disorders
Abstract
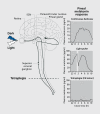
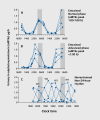
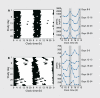
Many aspects of human physiology and behavior are dominated by 24-hour circadian rhythms that have a major impact on our health and well-being, including the sleep-wake cycle, alertness and performance patterns, and many daily hormone profiles. These rhythms are spontaneously generated by an internal "pacemaker" in the hypothalamus, and daily light exposure to the eyes is required to keep these circadian rhythms synchronized both internally and with the external environment. Sighted individuals take this daily synchronization process for granted, although they experience some of the consequences of circadian desynchrony when "jetlagged" or working night shifts. Most blind people with no perception of light, however, experience continual circadian desynchrony through a failure of light information to reach the hypothalamic circadian clock, resulting in cyclical episodes of poor sleep and daytime dysfunction. Daily melatonin administration, which provides a replacement synchronizing daily "time cue, " is a promising therapeutic strategy, although optimal treatment dose and timing remain to be determined.
Muchos aspectos fisiológicos y conductuales del ser humano están controlados por ritmos circadianos de 24 horas, los que tienen un gran impacto en nuestra salud y bienestar Estos ritmos incluyen el ciclo sueño-vigilia, el alerta y los patrones de varias funciones, como también el perfil de secreción diaria de muchas hormonas. Estos ritmos se generan espontáneamente por un “marcapaso” interno en el hipotálamo y se requiere de la exposición de los ojos a la luz diurna para mantener estos ritmos circadianos sincronizados tanto internamente como con el ambiente externo. Los sujetos videntes tienen este proceso de sincronización asegurado, aunque ellos experimenten algunas de las consecuencias derivadas de la falta de sincronía cuando tienen un “jetlag” o trabajan en turnos nocturnos. La mayoría de las personas ciegas no percibe la luz; sin embargo, experimentan continuas faltas de sincronía circadiana debido a que falla la llegada de la información luminosa al reloj circadiano hipotalámico, lo que se traduce en episodios cíclicos de mal dormir y mal funcionamiento durante el día. La administración diaria de melatonina proporciona un sustituto para la sincronización diaria de la “clave temporal”, y aunque constituye una estrategia terapéutica promisoria, aun falta determinar el momento más adecuado de la administración y la dosis terapéutica óptima.
De nombreux aspects de la physiologie et du comportement humain sont dominés par les rythmes circadiens de 24 h qui ont un impact majeur sur notre santé et notre bien-être, notamment le cycle veille-sommeil, les variations quotidiennes des processus attentionnels et de performance, et la sécrétion de nombreuses hormones. Ces rythmes circadiens sont générés spontanément par un « pacemaker » interne situé dans l'hypothalamus. Une exposition quotidienne des yeux à la lumière est nécessaire pour conserver la synchronisation de ces rythmes à la fois entre eux et avec l'environnement externe. Les voyants considèrent cette synchronisation quotidienne comme évidente, tout en expérimentant certaines conséquences de la désynchronisation circadienne en cas de décalage horaire ou de travail en horaire posté. La plupart des aveugles, n'ayant aucune perception de lumière, subissent une désynchronisation circadienne permanente par impossibilité de l'information lumineuse d'atteindre l'horloge circadienne hypothalamique. Ceci conduit à des épisodes cycliques de mauvais sommeil et de mauvais fonctionnement diurne. En resynchronisant les plages horaires chaque jour, l'administration quotidienne de mélatonine est une stratégie thérapeutique prometteuse, bien qu'il faille encore en préciser l'horaire d'administration et la posologie optimale.
Figures
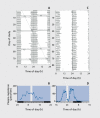
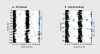
Similar articles
-
Non-24-Hour Sleep-Wake Rhythm Disorder in the Totally Blind: Diagnosis and Management.Front Neurol. 2017 Dec 18;8:686. doi: 10.3389/fneur.2017.00686. eCollection 2017. Front Neurol. 2017. PMID: 29326647 Free PMC article. Review.
-
Postoperative circadian disturbances.Dan Med Bull. 2010 Dec;57(12):B4205. Dan Med Bull. 2010. PMID: 21122464 Review.
-
Circadian Rhythm Sleep-Wake Disorders: a Contemporary Review of Neurobiology, Treatment, and Dysregulation in Neurodegenerative Disease.Neurotherapeutics. 2021 Jan;18(1):53-74. doi: 10.1007/s13311-021-01031-8. Epub 2021 Apr 12. Neurotherapeutics. 2021. PMID: 33844152 Free PMC article. Review.
-
Melatonin and the circadian system: contributions to successful female reproduction.Fertil Steril. 2014 Aug;102(2):321-8. doi: 10.1016/j.fertnstert.2014.06.014. Epub 2014 Jul 1. Fertil Steril. 2014. PMID: 24996495 Review.
-
The Effect of Blindness on Biological Rhythms and the Consequences of Circadian Rhythm Disorder.Turk J Ophthalmol. 2023 Apr 20;53(2):111-119. doi: 10.4274/tjo.galenos.2022.59296. Turk J Ophthalmol. 2023. PMID: 37089032 Free PMC article.
Cited by
-
Homeostatic and circadian contribution to EEG and molecular state variables of sleep regulation.Sleep. 2013 Mar 1;36(3):311-23. doi: 10.5665/sleep.2440. Sleep. 2013. PMID: 23450268 Free PMC article.
-
Study Protocol for Mindfulness-Based Yoga Versus Physical Exercise on the Psychological Well-Being in Students With Early Visual Impairment: A Three-Armed, Multi-Centered, Randomized Controlled Trial.Cureus. 2024 Sep 12;16(9):e69240. doi: 10.7759/cureus.69240. eCollection 2024 Sep. Cureus. 2024. PMID: 39398856 Free PMC article.
-
Caffeine does not entrain the circadian clock but improves daytime alertness in blind patients with non-24-hour rhythms.Sleep Med. 2015 Jun;16(6):800-4. doi: 10.1016/j.sleep.2015.01.018. Epub 2015 Feb 11. Sleep Med. 2015. PMID: 25891543 Free PMC article. Clinical Trial.
-
Impact of Circadian Rhythms on the Development and Clinical Management of Genitourinary Cancers.Front Oncol. 2022 Mar 9;12:759153. doi: 10.3389/fonc.2022.759153. eCollection 2022. Front Oncol. 2022. PMID: 35356228 Free PMC article. Review.
-
Protective role of melatonin in breast cancer: what we can learn from women with blindness.Cancer Causes Control. 2022 Jan;33(1):1-13. doi: 10.1007/s10552-021-01502-5. Epub 2021 Nov 27. Cancer Causes Control. 2022. PMID: 34837156 Review.
References
-
- Kneisley LW., Moskowitz MA., Lynch HJ. Cervical spinal cord lesions disrupt the rhythm in human melatonin excretion. J Neural Transm. 1 978;13(suppl):311–323. - PubMed
-
- Zeitzer JM., Ayas NT., Shea SA., Brown R., Czeisler CA. Absence of detectable melatonin and perservation of Cortisol and thyrotropin rhythms in tetraplegia. J Clin Endocrinol Metab. 2000;85:2189–2196. - PubMed
-
- Wehr TA., Duncan WC. Jr., Sher L., et al. A circadian signal of change of season in patients with seasonal affective disorder. Arch Gen Psychiatry. 2001;58:1108–1114. - PubMed
-
- Zeitzer JM., Ayas NT., Shea SA., Brown R. Czeisler CA Absence of detectable melatonin and preservation of Cortisol and thyrotropin rhythms in tetraplegia. J Clin Endocrinol Metab. 2000;85:2189–2196. - PubMed
-
- Saper CB., Scammell TE., Lu J. Hypothalamic regulation of sleep and circadian rhythms. Nature. 2005;437:1257–1263. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
