The TR2 and TR4 orphan nuclear receptors repress Gata1 transcription
- PMID: 17974920
- PMCID: PMC2045135
- DOI: 10.1101/gad.1593307
The TR2 and TR4 orphan nuclear receptors repress Gata1 transcription
Abstract
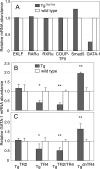
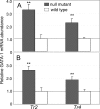
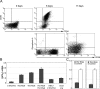
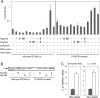
When the orphan nuclear receptors TR2 and TR4, the DNA-binding subunits of the DRED repressor complex, are forcibly expressed in erythroid cells of transgenic mice, embryos exhibit a transient mid-gestational anemia as a consequence of a reduction in the number of primitive erythroid cells. GATA-1 mRNA is specifically diminished in the erythroid cells of these TR2/TR4 transgenic embryos as it is in human CD34(+) progenitor cells transfected with forcibly expressed TR2/TR4. In contrast, in loss-of-function studies analyzing either Tr2- or Tr4-germline-null mutant mice or human CD34(+) progenitor cells transfected with force-expressed TR2 and TR4 short hairpin RNAs (shRNAs), GATA-1 mRNA is induced. An evolutionarily conserved direct repeat (DR) element, a canonical binding site for nuclear receptors, was identified in the GATA1 hematopoietic enhancer (G1HE), and TR2/TR4 binds to that site in vitro and in vivo. Mutation of that DR element led to elevated Gata1 promoter activity, and reduced promoter responsiveness to cotransfected TR2/TR4. Thus, TR2/TR4 directly represses Gata1/GATA1 transcription in murine and human erythroid progenitor cells through an evolutionarily conserved binding site within a well-characterized, tissue-specific Gata1 enhancer, thereby providing a mechanism by which Gata1 can be directly silenced during terminal erythroid maturation.
Figures


References
-
- Anguita E., Hughes J., Heyworth C., Blobel G.A., Wood W.G., Higgs D.R., Hughes J., Heyworth C., Blobel G.A., Wood W.G., Higgs D.R., Heyworth C., Blobel G.A., Wood W.G., Higgs D.R., Blobel G.A., Wood W.G., Higgs D.R., Wood W.G., Higgs D.R., Higgs D.R. Globin gene activation during haemopoiesis is driven by protein complexes nucleated by GATA-1 and GATA-2. EMBO J. 2004;23:2841–2852. - PMC - PubMed
-
- Baines P., Mayani H., Bains M., Fisher J., Hoy T., Jacobs A., Mayani H., Bains M., Fisher J., Hoy T., Jacobs A., Bains M., Fisher J., Hoy T., Jacobs A., Fisher J., Hoy T., Jacobs A., Hoy T., Jacobs A., Jacobs A. Enrichment of CD34 (My10)-positive myeloid and erythroid progenitors from human marrow and their growth in cultures supplemented with recombinant human granulocyte-macrophage colony-stimulating factor. Exp. Hematol. 1988;16:785–789. - PubMed
-
- Collins S.J. The role of retinoids and retinoic acid receptors in normal hematopoiesis. Leukemia. 2002;16:1896–1905. - PubMed
-
- Collins L.L., Lee Y.F., Heinlein C.A., Liu N.C., Chen Y.T., Shyr C.R., Meshul C.K., Uno H., Platt K.A., Chang C., Lee Y.F., Heinlein C.A., Liu N.C., Chen Y.T., Shyr C.R., Meshul C.K., Uno H., Platt K.A., Chang C., Heinlein C.A., Liu N.C., Chen Y.T., Shyr C.R., Meshul C.K., Uno H., Platt K.A., Chang C., Liu N.C., Chen Y.T., Shyr C.R., Meshul C.K., Uno H., Platt K.A., Chang C., Chen Y.T., Shyr C.R., Meshul C.K., Uno H., Platt K.A., Chang C., Shyr C.R., Meshul C.K., Uno H., Platt K.A., Chang C., Meshul C.K., Uno H., Platt K.A., Chang C., Uno H., Platt K.A., Chang C., Platt K.A., Chang C., Chang C. Growth retardation and abnormal maternal behavior in mice lacking testicular orphan nuclear receptor 4. Proc. Natl. Acad. Sci. 2004;101:15058–15063. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
