The saga of MHC-bound peptides: a renaissance for antigen presentation?
- PMID: 17975658
- PMCID: PMC2045625
- DOI: 10.1172/JCI33997
The saga of MHC-bound peptides: a renaissance for antigen presentation?
Abstract
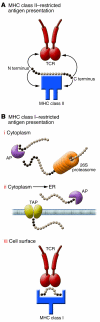
In this issue of the JCI, two separate studies on MHC-bound peptides reopen the debate on the utility of peptides for the purposes of vaccination and treatment of autoimmune diseases. In the first study, by Wahlström et al., peptides bound to HLA-DR17 from bronchoalveolar lavage cells of sarcoidosis patients were analyzed in order to identify target antigens of the autoimmune response (see the related article beginning on page 3576). In the second study, by Le Gall et al., the modulation of epitope immunodominance and the processing and presentation of HIV peptides for MHC class I recognition were shown to be dependent on flanking residues that were N terminal to the natural epitopes (see the related article beginning on page 3563). Both studies highlight the tremendous therapeutic potential of MHC-bound peptides. They also emphasize that technical issues are still plaguing this field and hindering our understanding of MHC presentation in vivo.
Figures
Comment on
-
Portable flanking sequences modulate CTL epitope processing.J Clin Invest. 2007 Nov;117(11):3563-75. doi: 10.1172/JCI32047. J Clin Invest. 2007. PMID: 17975674 Free PMC article.
-
Identification of HLA-DR-bound peptides presented by human bronchoalveolar lavage cells in sarcoidosis.J Clin Invest. 2007 Nov;117(11):3576-82. doi: 10.1172/JCI32401. J Clin Invest. 2007. PMID: 17975675 Free PMC article.
Similar articles
-
Approach for Identifying Human Leukocyte Antigen (HLA)-DR Bound Peptides from Scarce Clinical Samples.Mol Cell Proteomics. 2016 Sep;15(9):3017-29. doi: 10.1074/mcp.M116.060764. Epub 2016 Jul 24. Mol Cell Proteomics. 2016. PMID: 27452731 Free PMC article.
-
Recognition of core and flanking amino acids of MHC class II-bound peptides by the T cell receptor.Eur J Immunol. 2002 Sep;32(9):2510-20. doi: 10.1002/1521-4141(200209)32:9<2510::AID-IMMU2510>3.0.CO;2-Q. Eur J Immunol. 2002. PMID: 12207335
-
Presentation and recognition of major and minor histocompatibility antigens.Transpl Immunol. 1994 Jun;2(2):103-7. doi: 10.1016/0966-3274(94)90036-1. Transpl Immunol. 1994. PMID: 7953301 Review.
-
Peptides presented in vivo by HLA-DR in thyroid autoimmunity.Adv Immunol. 2008;99:165-209. doi: 10.1016/S0065-2776(08)00606-8. Adv Immunol. 2008. PMID: 19117535 Review.
-
Endogenous presentation of a nascent antigenic epitope to CD8+ CTL is more efficient than exogenous presentation.Immunol Cell Biol. 1996 Oct;74(5):394-400. doi: 10.1038/icb.1996.68. Immunol Cell Biol. 1996. PMID: 8912001
Cited by
-
HDX-ESI-MS reveals enhanced conformational dynamics of the amyloidogenic protein beta(2)-microglobulin upon release from the MHC-1.J Am Soc Mass Spectrom. 2009 Feb;20(2):278-86. doi: 10.1016/j.jasms.2008.10.005. Epub 2008 Oct 17. J Am Soc Mass Spectrom. 2009. PMID: 18996721 Free PMC article.
-
Peptides presented by HLA-DR molecules in synovia of patients with rheumatoid arthritis or antibiotic-refractory Lyme arthritis.Mol Cell Proteomics. 2011 Mar;10(3):M110.002477. doi: 10.1074/mcp.M110.002477. Epub 2010 Nov 16. Mol Cell Proteomics. 2011. PMID: 21081667 Free PMC article.
-
Identifying a Small Molecule Blocking Antigen Presentation in Autoimmune Thyroiditis.J Biol Chem. 2016 Feb 19;291(8):4079-90. doi: 10.1074/jbc.M115.694687. Epub 2015 Dec 24. J Biol Chem. 2016. PMID: 26703475 Free PMC article.
-
Retro-inverso D-peptides as a novel targeted immunotherapy for Type 1 diabetes.J Autoimmun. 2020 Dec;115:102543. doi: 10.1016/j.jaut.2020.102543. Epub 2020 Sep 17. J Autoimmun. 2020. PMID: 32951964 Free PMC article.
References
-
- Bjorkman P.J., et al. Structure of the human class I histocompatibility antigen, HLA-A2. Nature. 1987;329:506–512. - PubMed
-
- Townsend A.R., et al. The epitopes of influenza nucleoprotein recognized by cytotoxic T lymphocytes can be defined with short synthetic peptides. Cell. 1986;44:959–968. - PubMed
-
- Berlin M., Fogdell-Hahn A., Olerup O., Eklund A., Grunewald J. HLA-DR predicts the prognosis in Scandinavian patients with pulmonary sarcoidosis. Am. J. Respir. Crit. Care Med. 1997;156:1601–1605. - PubMed
-
- Katchar K., Wahlstrom J., Eklund A., Grunewald J. Highly activated T-cell receptor AV2S3(+) CD4(+) lung T-cell expansions in pulmonary sarcoidosis. Am. J. Respir. Crit. Care Med. 2001;163:1540–1545. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

