Differential expression of matrix metalloproteinases during stimulated ovarian recrudescence in Siberian hamsters (Phodopus sungorus)
- PMID: 17980368
- PMCID: PMC2262402
- DOI: 10.1016/j.ygcen.2007.09.003
Differential expression of matrix metalloproteinases during stimulated ovarian recrudescence in Siberian hamsters (Phodopus sungorus)
Abstract
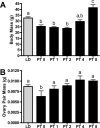
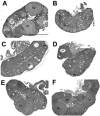
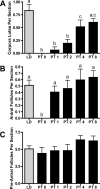
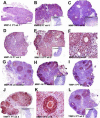
The matrix metalloproteinases (MMPs) are a family of extracellular matrix-cleaving enzymes involved in ovarian remodeling. In many non-tropical species, including Siberian hamsters, ovarian remodeling is necessary for the functional changes associated with seasonal reproduction. We evaluated MMPs and their endogenous inhibitors (TIMPs), during photoperiod-induced ovarian recrudescence in Siberian hamsters. Hamsters were transferred from long day (LD; 16:8) to short day (SD; 8:16) photoperiods for 14weeks, and then returned to LD for 0, 1, 2, 4, or 8weeks for collection of ovaries and plasma. Post-transfer (PT) LD exposure increased body and ovarian mass. Number of corpora lutea and antral, but not preantral follicles increased in PT groups. Plasma estradiol concentrations were lower in PT weeks 0-4, and returned to LD levels at PT week 8. No change was observed in relative MMP/TIMP mRNA levels at PT week 0 (SD week 14) as compared to LD. Photostimulation increased MMP-2 mRNA at PT week 8 as compared to PT weeks 0-1. MMP-14 mRNA expression peaked at PT weeks 1-2 as compared to LD levels, while MMP-13 expression was low during this time. TIMP-1 mRNA peaked at PT week 8 as compared to PT weeks 0-4. No changes were noted in MMP-9 and TIMP-2 mRNA expression. In general, MMP/TIMP protein immunodetection followed the same patterns with most staining occurring in granulosa cells of follicles and corpora lutea. Our data suggest that mRNA and protein for several members of the MMP/TIMP families are expressed in Siberian hamster ovaries during recrudescence. Because of the variation observed in expression patterns, MMPs and TIMPs may be differentially involved with photostimulated return to ovarian function.
Figures




Similar articles
-
Differential activity of matrix metalloproteinases (MMPs) during photoperiod induced uterine regression and recrudescence in Siberian hamsters (Phodopus sungorus).Mol Reprod Dev. 2008 Sep;75(9):1433-40. doi: 10.1002/mrd.20879. Mol Reprod Dev. 2008. PMID: 18213647 Free PMC article.
-
Ovarian matrix metalloproteinases are differentially regulated during the estrous cycle but not during short photoperiod induced regression in Siberian hamsters (Phodopus sungorus).Reprod Biol Endocrinol. 2010 Jun 25;8:79. doi: 10.1186/1477-7827-8-79. Reprod Biol Endocrinol. 2010. PMID: 20579366 Free PMC article.
-
Inhibition of matrix metalloproteinases in Siberian hamsters impedes photostimulated recrudescence of ovaries.Reproduction. 2010 Dec;140(6):875-83. doi: 10.1530/REP-10-0304. Epub 2010 Sep 29. Reproduction. 2010. PMID: 20881024 Free PMC article.
-
MMPS and TIMPS in ovarian physiology and pathophysiology.Front Biosci. 2004 Sep 1;9:2474-83. doi: 10.2741/1409. Front Biosci. 2004. PMID: 15353300 Review.
-
The regulation of matrix metalloproteinases and their inhibitors.Int J Biochem Cell Biol. 2008;40(6-7):1362-78. doi: 10.1016/j.biocel.2007.12.006. Epub 2007 Dec 24. Int J Biochem Cell Biol. 2008. PMID: 18258475 Review.
Cited by
-
Assessing recrudescence of photoregressed Siberian hamster ovaries using in vitro whole ovary culture.Mol Reprod Dev. 2018 Oct;85(10):746-759. doi: 10.1002/mrd.23050. Epub 2018 Sep 7. Mol Reprod Dev. 2018. PMID: 30091812 Free PMC article.
-
Effect of PACAP/PAC1R on Follicle Development of Djungarian Hamster (Phodopus sungorus) with the Variation of Ambient Temperatures.Biology (Basel). 2023 Feb 15;12(2):315. doi: 10.3390/biology12020315. Biology (Basel). 2023. PMID: 36829590 Free PMC article.
-
Photoperiod-driven testicular DNA methylation in gonadotropin and sex steroid receptor promoters in Siberian hamsters.J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2025 May;211(3):327-337. doi: 10.1007/s00359-025-01733-w. Epub 2025 Feb 15. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2025. PMID: 39954063 Free PMC article.
-
Differential activity of matrix metalloproteinases (MMPs) during photoperiod induced uterine regression and recrudescence in Siberian hamsters (Phodopus sungorus).Mol Reprod Dev. 2008 Sep;75(9):1433-40. doi: 10.1002/mrd.20879. Mol Reprod Dev. 2008. PMID: 18213647 Free PMC article.
-
Folliculogenic factors in photoregressed ovaries: Differences in mRNA expression in early compared to late follicle development.Gen Comp Endocrinol. 2018 May 1;260:90-99. doi: 10.1016/j.ygcen.2018.01.003. Epub 2018 Jan 6. Gen Comp Endocrinol. 2018. PMID: 29317212 Free PMC article.
References
-
- Bakke LJ, Dow MP, Cassar CA, Peters MW, Pursley JR, Smith GW. Effect of the preovulatory gonadotropin surge on matrix metalloproteinase (MMP)-14, MMP-2, and tissue inhibitor of metalloproteinases-2 expression within bovine periovulatory follicular and luteal tissue. Biol Reprod. 2002;66:1627–34. - PubMed
-
- Bakke LJ, Li Q, Cassar CA, Dow MP, Pursley JR, Smith GW. Gonadotropin surge-induced differential upregulation of collagenase-1 (MMP-1) and collagenase-3 (MMP-13) mRNA and protein in bovine preovulatory follicles. Biol Reprod. 2004;71:605–12. - PubMed
-
- Brännström M, Woessner JF, Koos RD, Sear CH, LeMaire WJ. Inhibitors of mammalian tissue collagenase and metalloproteinases suppress ovulation in the perfused rat ovary. Endocrinology. 1988;122:1715–1721. - PubMed
-
- Butler TA, Zhu C, Mueller RA, Fuller GC, Lemaire WJ, Woessner JF. Inhibition of ovulation in the perfused rat ovary by the synthetic collagenase inhibitor SC 44463. Biol. Reprod. 1991;44:1183–1188. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

