beta-arrestin 2 oligomerization controls the Mdm2-dependent inhibition of p53
- PMID: 17984062
- PMCID: PMC2084296
- DOI: 10.1073/pnas.0705550104
beta-arrestin 2 oligomerization controls the Mdm2-dependent inhibition of p53
Abstract
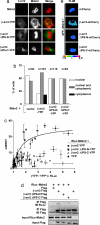
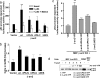
beta-arrestins (beta-arrs), two ubiquitous proteins involved in serpentine heptahelical receptor regulation and signaling, form constitutive homo- and heterooligomers stabilized by inositol 1,2,3,4,5,6-hexakisphosphate (IP6). Monomeric beta-arrs are believed to interact with receptors after agonist activation, and therefore, beta-arr oligomers have been proposed to represent a resting biologically inactive state. In contrast to this, we report here that the interaction with and subsequent titration out of the nucleus of the protooncogene Mdm2 specifically require beta-arr2 oligomers together with the previously characterized nucleocytoplasmic shuttling of beta-arr2. Mutation of the IP6-binding sites impair oligomerization, reduce interaction with Mdm2, and inhibit p53-dependent antiproliferative effects of beta-arr2, whereas the competence for receptor regulation and signaling is maintained. These observations suggest that the intracellular concentration of beta-arr2 oligomers might control cell survival and proliferation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Lohse MJ, Benovic JL, Codina J, Caron MG, Lefkowitz RJ. Science. 1990;248:1547–1550. - PubMed
-
- Ferguson SS, Downey WE, Colapietro AM, Barak LS, Menard L, Caron MG. Science. 1996;271:363–366. - PubMed
-
- Goodman OBJ, Krupnick JG, Santini F, Gurevich VV, Penn RB, Gagnon AW, Keen JH, Benovic JL. Nature. 1996;383:447–450. - PubMed
-
- Shenoy SK, Lefkowitz RJ. Sci STKE. 2005;2005:cm10. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

