Modulation of intestinal goblet cell function during infection by an attaching and effacing bacterial pathogen
- PMID: 17984203
- PMCID: PMC2223480
- DOI: 10.1128/IAI.00093-07
Modulation of intestinal goblet cell function during infection by an attaching and effacing bacterial pathogen
Abstract
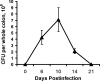
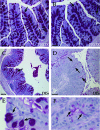
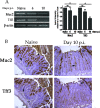
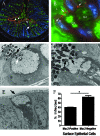
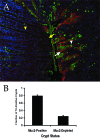
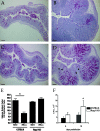
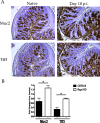
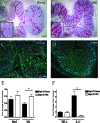
The attaching and effacing (A/E) bacterial pathogens enteropathogenic Escherichia coli and enterohemorrhagic E. coli and the related mouse pathogen Citrobacter rodentium colonize their hosts' intestines by infecting the apical surfaces of enterocytes, subverting their function, and they ultimately cause diarrhea. Surprisingly, little is known about the interactions of these organisms with goblet cells, which are specialized epithelial cells that secrete the protective molecules Muc2 and trefoil factor 3 (Tff3) into the intestinal lumen. C. rodentium infection leads to dramatic goblet cell depletion within the infected colon, yet it is not clear whether C. rodentium infects goblet cells or if this pathology is pathogen or host mediated. As determined by immunostaining and PCR, both the number of goblet cells and the expression of genes encoding Muc2 and Tff3 were significantly reduced by day 10 postinfection. While electron microscopy and immunostaining revealed that C. rodentium directly infected a fraction of colonic goblet cells, C. rodentium localization did not correlate with goblet cell depletion. To assess the role of the host immune system in these changes, Rag1 knockout (KO) (T- and B-cell-deficient) mice were infected with C. rodentium. Rag1 KO mice did not exhibit the reduction in the number of goblet cells or in mediator (Muc2 and Tff3) expression observed in infected immunocompetent mice. However, reconstitution of Rag1 KO mice with T and B lymphocytes from C57BL/6 mice restored the goblet cell depletion phenotype during C. rodentium infection. In conclusion, these studies demonstrated that while colonic goblet cells can be subject to direct infection and potential subversion by A/E pathogens in vivo, it is the host immune system that primarily modulates the function of these cells during infection.
Figures

References
-
- Arnold, J. W., G. R. Klimpel, and D. W. Niesel. 1993. Tumor necrosis factor (TNFα) regulates intestinal mucus production during salmonellosis. Cell. Immunol. 151336-344. - PubMed
-
- Artis, D., C. S. Potten, K. J. Else, F. D. Finkelman, and R. K. Grencis. 1999. Trichuris muris: host intestinal epithelial cell hyperproliferation during chronic infection is regulated by interferon-gamma. Exp. Parasitol. 92144-153. - PubMed
-
- Artis, D., M. L. Wang, S. A. Keilbaugh, W. He, M. Brenes, G. P. Swain, P. A. Knight, D. D. Donaldson, M. A. Lazar, H. R. P. Miller, G. A. Schad, P. Scott, and G. D. Wu. 2004. RELMβ/FIZZ2 is a goblet cell-specific immune-effector molecule in the gastrointestinal tract Proc. Natl. Acad. Sci. USA 10113596-13600. - PMC - PubMed
-
- Boyle, E. C., N. F. Brown, and B. B. Finlay. 2006. Salmonella enterica serovar Typhimurium effectors SopB, SopE, SopE2 and SipA disrupt tight junction structure and function. Cell. Microbiol. 81946-1957. - PubMed
-
- Brand, S., F. Beigel, T. Olszak, K. Zitzmann, S. T. Eichhorst, J.-M. Otte, J. Diebold, H. Diepolder, B. Adler, C. J. Auernhammer, B. Goke, and J. Dambacher. 2005. IL-28A and IL-29 mediate antiproliferative and antiviral signals in intestinal epithelial cells and murine CMV infection increases colonic IL-28A expression. Am. J. Physiol. Gastrointest. Liver Physiol. 289G960-G968. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

