In vivo methods for the assessment of topical drug bioavailability
- PMID: 17985216
- PMCID: PMC2217624
- DOI: 10.1007/s11095-007-9429-7
In vivo methods for the assessment of topical drug bioavailability
Abstract
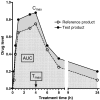
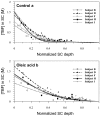
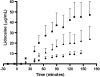
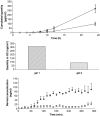
This paper reviews some current methods for the in vivo assessment of local cutaneous bioavailability in humans after topical drug application. After an introduction discussing the importance of local drug bioavailability assessment and the limitations of model-based predictions, the focus turns to the relevance of experimental studies. The available techniques are then reviewed in detail, with particular emphasis on the tape stripping and microdialysis methodologies. Other less developed techniques, including the skin biopsy, suction blister, follicle removal and confocal Raman spectroscopy techniques are also described.
Figures







References
-
- None
- A. Finlay, and R. Marks. Determination of corticosteroid concentration profile in the stratum corneum using the skin surface biopsy technique. Br. J. Derm.107Suppl 22:23 (1982).
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'PubMed', 'value': '20479733', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/20479733/'}]}
- P. M. Elias. Epidermal lipids, barrier function and desquamation. J. Invest. Dermatol.80:S44–S49 (1983). - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'PubMed', 'value': '2746002', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/2746002/'}]}
- P. W. Wertz, D. C. Swartzendruber, D. J. Kitko, K. C. Madison, and D. T. Downing. The role of the corneocytes lipid envelopes in cohesion of the stratum corneum. J. Invest. Dermatol.93:169–172 (1989). - PubMed
-
- None
- P. M. Elias. Epidermal barrier function: intercellular lamellar lipid structures, origin, composition and metabolism. J. Control. Release15:199–208 (1991).
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'PubMed', 'value': '4718753', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/4718753/'}]}
- R. L. Anderson, and J. M. Cassidy. Variations in physical dimensions and chemical composition of human stratum corneum. J. Invest. Dermatol.61:30–32 (1973). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

