Activity-dependent heteromerization of the hyperpolarization-activated, cyclic-nucleotide gated (HCN) channels: role of N-linked glycosylation
- PMID: 17988239
- PMCID: PMC2747799
- DOI: 10.1111/j.1471-4159.2007.05110.x
Activity-dependent heteromerization of the hyperpolarization-activated, cyclic-nucleotide gated (HCN) channels: role of N-linked glycosylation
Abstract
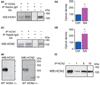
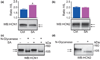
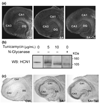
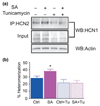
Formation of heteromeric complexes of ion channels via co-assembly of different subunit isoforms provides an important mechanism for enhanced channel diversity. We have previously demonstrated co-association of the hyperpolarization activated cyclic-nucleotide gated (HCN1/HCN2) channel isoforms that was regulated by network (seizure) activity in developing hippocampus. However, the mechanisms that underlie this augmented expression of heteromeric complexes have remained unknown. Glycosylation of the HCN channels has been implicated in the stabilization and membrane expression of heteromeric HCN1/HCN2 constructs in heterologous systems. Therefore, we used in vivo and in vitro systems to test the hypothesis that activity modifies HCN1/HCN2 heteromerization in neurons by modulating the glycosylation state of the channel molecules. Seizure-like activity (SA) increased HCN1/HCN2 heteromerization in hippocampus in vivo as well as in hippocampal organotypic slice cultures. This activity increased the abundance of glycosylated HCN1 but not HCN2-channel molecules. In addition, glycosylated HCN1 channels were preferentially co-immunoprecipitated with the HCN2 isoforms. Provoking SA in vitro in the presence of the N-linked glycosylation blocker tunicamycin abrogated the activity-dependent increase of HCN1/HCN2 heteromerization. Thus, hippocampal HCN1 molecules have a significantly higher probability of being glycosylated after SA, and this might promote a stable heteromerization with HCN2.
Figures


Similar articles
-
Role of subunit heteromerization and N-linked glycosylation in the formation of functional hyperpolarization-activated cyclic nucleotide-gated channels.J Biol Chem. 2003 Oct 31;278(44):43781-6. doi: 10.1074/jbc.M306958200. Epub 2003 Aug 19. J Biol Chem. 2003. PMID: 12928435
-
Properties of hyperpolarization-activated pacemaker current defined by coassembly of HCN1 and HCN2 subunits and basal modulation by cyclic nucleotide.J Gen Physiol. 2001 May;117(5):491-504. doi: 10.1085/jgp.117.5.491. J Gen Physiol. 2001. PMID: 11331358 Free PMC article.
-
Formation of heteromeric hyperpolarization-activated cyclic nucleotide-gated (HCN) channels in the hippocampus is regulated by developmental seizures.Neurobiol Dis. 2005 Jun-Jul;19(1-2):200-7. doi: 10.1016/j.nbd.2004.12.015. Neurobiol Dis. 2005. PMID: 15837575 Free PMC article.
-
Voltage sensing and activation gating of HCN pacemaker channels.Trends Cardiovasc Med. 2002 Jan;12(1):42-5. doi: 10.1016/s1050-1738(01)00141-4. Trends Cardiovasc Med. 2002. PMID: 11796244 Review.
-
CNG and HCN channels: two peas, one pod.Annu Rev Physiol. 2006;68:375-401. doi: 10.1146/annurev.physiol.68.040104.134728. Annu Rev Physiol. 2006. PMID: 16460277 Review.
Cited by
-
Double Trouble? Potential for Hyperexcitability Following Both Channelopathic up- and Downregulation of I(h) in Epilepsy.Front Neurosci. 2009 May 1;3(1):25-33. doi: 10.3389/neuro.01.005.2009. eCollection 2009 May. Front Neurosci. 2009. PMID: 19753094 Free PMC article.
-
The Contribution of HCN Channelopathies in Different Epileptic Syndromes, Mechanisms, Modulators, and Potential Treatment Targets: A Systematic Review.Front Mol Neurosci. 2022 May 19;15:807202. doi: 10.3389/fnmol.2022.807202. eCollection 2022. Front Mol Neurosci. 2022. PMID: 35663267 Free PMC article.
-
Immunolocalization of hyperpolarization-activated cationic HCN1 and HCN3 channels in the rat nephron: regulation of HCN3 by potassium diets.Histochem Cell Biol. 2016 Jan;145(1):25-40. doi: 10.1007/s00418-015-1375-6. Epub 2015 Oct 29. Histochem Cell Biol. 2016. PMID: 26515056
-
Nucleus accumbens hyperpolarization-activated cyclic nucleotide-gated channels modulate methamphetamine self-administration in rats.Psychopharmacology (Berl). 2016 Aug;233(15-16):3017-29. doi: 10.1007/s00213-016-4349-z. Epub 2016 Jun 22. Psychopharmacology (Berl). 2016. PMID: 27329413
-
Distinct perinatal features of the hyperpolarization-activated non-selective cation current I(h) in the rat cortical plate.Neural Dev. 2012 Jun 13;7:21. doi: 10.1186/1749-8104-7-21. Neural Dev. 2012. PMID: 22694806 Free PMC article.
References
-
- Bräuer AU, Savaskan NE, Kole MH, et al. Molecular and functional analysis of hyperpolarization-activated pacemaker channels in the hippocampus after entorhinal cortex lesion. FASEB J. 2001;15:2689–2701. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

