The transcription factor aryl hydrocarbon receptor nuclear translocator functions as an estrogen receptor beta-selective coactivator, and its recruitment to alternative pathways mediates antiestrogenic effects of dioxin
- PMID: 17991765
- PMCID: PMC5419643
- DOI: 10.1210/me.2007-0128
The transcription factor aryl hydrocarbon receptor nuclear translocator functions as an estrogen receptor beta-selective coactivator, and its recruitment to alternative pathways mediates antiestrogenic effects of dioxin
Abstract
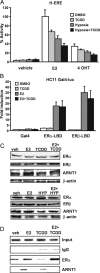
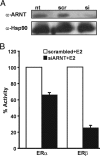
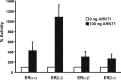
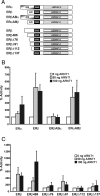
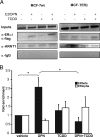
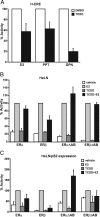
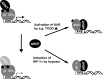
The biological effects of dioxins are mediated by the aryl hydrocarbon receptor (AhR) and its dimerization partner, the AhR nuclear translocator (ARNT), and include interference with hormonal signaling pathways like the response to estrogens. The effects of estrogens are mediated by two estrogen receptor (ER) isoforms, ERalpha and ERbeta, which belong to the family of nuclear receptors. We have previously shown that ARNT can act as coactivator of the ERs. In this study, we show that recruitment of ARNT to AhR or hypoxia-inducible factor-1alpha signaling pathways as well as small interfering RNA-mediated down-regulation of ARNT levels lead to a reduction in ER transcriptional activity. Using chromatin immunoprecipitation assays, we demonstrate that this decrease coincides with reduced recruitment of ARNT to estradiol-regulated promoters. We show further that coactivation by ARNT as well as inhibition by dioxin acts stronger on ERbeta than on ERalpha activity. Additionally, we demonstrate that the effects of ARNT are dependent on the A/B domain of the ERs with the A/B domain of ERbeta being considerably stronger in mediating the coactivating effects of ARNT. Taken together, our studies show that recruitment of ARNT to the AhR after dioxin treatment can account for the antiestrogenic effect of dioxins. Moreover, we show for the first time that the inhibitory effects of dioxin are more pronounced on ERbeta than on ERalpha.
Figures

Similar articles
-
Hypoxia perturbs aryl hydrocarbon receptor signaling and CYP1A1 expression induced by PCB 126 in human skin and liver-derived cell lines.Toxicol Appl Pharmacol. 2014 Feb 1;274(3):408-16. doi: 10.1016/j.taap.2013.12.002. Epub 2013 Dec 16. Toxicol Appl Pharmacol. 2014. PMID: 24355420 Free PMC article.
-
Distinct roles for aryl hydrocarbon receptor nuclear translocator and ah receptor in estrogen-mediated signaling in human cancer cell lines.PLoS One. 2012;7(1):e29545. doi: 10.1371/journal.pone.0029545. Epub 2012 Jan 3. PLoS One. 2012. PMID: 22235307 Free PMC article.
-
ER alpha-AHR-ARNT protein-protein interactions mediate estradiol-dependent transrepression of dioxin-inducible gene transcription.J Biol Chem. 2005 Jun 3;280(22):21607-11. doi: 10.1074/jbc.C500090200. Epub 2005 Apr 18. J Biol Chem. 2005. PMID: 15837795
-
Mechanisms of inhibitory aryl hydrocarbon receptor-estrogen receptor crosstalk in human breast cancer cells.J Mammary Gland Biol Neoplasia. 2000 Jul;5(3):295-306. doi: 10.1023/a:1009550912337. J Mammary Gland Biol Neoplasia. 2000. PMID: 14973392 Review.
-
Regulatory crosstalk and interference between the xenobiotic and hypoxia sensing pathways at the AhR-ARNT-HIF1α signaling node.Chem Biol Interact. 2014 Jul 25;218:82-8. doi: 10.1016/j.cbi.2014.05.001. Epub 2014 May 10. Chem Biol Interact. 2014. PMID: 24824450 Free PMC article. Review.
Cited by
-
A model for aryl hydrocarbon receptor-activated gene expression shows potency and efficacy changes and predicts squelching due to competition for transcription co-activators.PLoS One. 2015 Jun 3;10(6):e0127952. doi: 10.1371/journal.pone.0127952. eCollection 2015. PLoS One. 2015. PMID: 26039703 Free PMC article.
-
The aryl hydrocarbon receptor nuclear translocator-interacting protein 2 suppresses the estrogen receptor signaling via an Arnt-dependent mechanism.Arch Biochem Biophys. 2010 Oct 15;502(2):121-9. doi: 10.1016/j.abb.2010.07.022. Epub 2010 Jul 29. Arch Biochem Biophys. 2010. PMID: 20674540 Free PMC article.
-
Genetic variability in glutamic acid decarboxylase genes: associations with post-traumatic seizures after severe TBI.Epilepsy Res. 2013 Feb;103(2-3):180-94. doi: 10.1016/j.eplepsyres.2012.07.006. Epub 2012 Jul 26. Epilepsy Res. 2013. PMID: 22840783 Free PMC article. Clinical Trial.
-
Mechanisms of circadian clock interactions with aryl hydrocarbon receptor signalling.Eur J Neurosci. 2020 Jan;51(1):379-395. doi: 10.1111/ejn.14361. Epub 2019 Feb 25. Eur J Neurosci. 2020. PMID: 30706546 Free PMC article.
-
Coactivator recruitment of AhR/ARNT1.Int J Mol Sci. 2014 Jun 19;15(6):11100-10. doi: 10.3390/ijms150611100. Int J Mol Sci. 2014. PMID: 24950180 Free PMC article. Review.
References
-
- Nilsson S, Gustafsson JA 2002. Biological role of estrogen and estrogen receptors. Crit Rev Biochem Mol Biol 37:1–28 - PubMed
-
- Gronemeyer H, Gustafsson JA, Laudet V 2004. Principles for modulation of the nuclear receptor superfamily. Nat Rev Drug Discov 3:950–964 - PubMed
-
- Nilsson S, Makela S, Treuter E, Tujague M, Thomsen J, Andersson G, Enmark E, Pettersson K, Warner M, Gustafsson JA 2001. Mechanisms of estrogen action. Physiol Rev 81:1535–1565 - PubMed
-
- Pettersson K, Gustafsson JA 2001. Role of estrogen receptor β in estrogen action. Annu Rev Physiol 63:165–192 - PubMed
-
- Poland A, Knutson JC 1982. 2,3,7,8-Tetrachlorodibenzo-p-dioxin and related halogenated aromatic hydrocarbons: examination of the mechanism of toxicity. Annu Rev Pharmacol Toxicol 22:517–554 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

