Anticancer activity of oncolytic adenovirus vector armed with IFN-alpha and ADP is enhanced by pharmacologically controlled expression of TRAIL
- PMID: 17992200
- PMCID: PMC3433948
- DOI: 10.1038/sj.cgt.7701107
Anticancer activity of oncolytic adenovirus vector armed with IFN-alpha and ADP is enhanced by pharmacologically controlled expression of TRAIL
Abstract
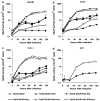
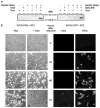
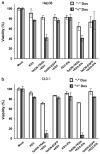
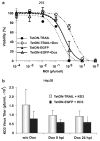
We have previously described oncolytic adenovirus (Ad) vectors KD3 and KD3-interferon (IFN) that were rendered cancer-specific by mutations in the E1A region of Ad; these mutations abolish binding of E1A proteins to p300/CBP and pRB. The antitumor activity of the vectors was enhanced by overexpression of the Adenovirus Death Protein (ADP, E3-11.6K) and by replication-linked expression of IFN-alpha. We hypothesized that the anticancer efficacy of the KD3-IFN vector could be further improved by expression of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL). E1-deleted Ad vectors were constructed carrying reporter genes for enhanced green fluorescent protein or secreted placental alkaline phosphatase (SEAP) and a therapeutic gene for TRAIL under control of the TetON system. Expression of the genes was increased in the presence of a helper virus and the inducer doxycycline such that up to 231-fold activation of expression for the TetON-SEAP vector was obtained. Coinfection with TetON-TRAIL augmented oncolytic activity of KD3 and KD3-IFN in vitro. Induction of TRAIL expression did not reduce the yield of progeny virus. Combination of TetON-TRAIL and KD3-IFN produced superior antitumor activity in vivo as compared with either vector alone demonstrating the efficacy of a four-pronged cancer gene therapy approach, which includes Ad oncolysis, ADP overexpression, IFN-alpha-mediated immunotherapy, and pharmacologically controlled TRAIL activity.
Figures

Similar articles
-
Targeting interferon-alpha increases antitumor efficacy and reduces hepatotoxicity of E1A-mutated spread-enhanced oncolytic adenovirus.Mol Ther. 2007 Mar;15(3):598-607. doi: 10.1038/sj.mt.6300064. Epub 2006 Dec 26. Mol Ther. 2007. PMID: 17191072
-
Radiation increases the activity of oncolytic adenovirus cancer gene therapy vectors that overexpress the ADP (E3-11.6K) protein.Cancer Gene Ther. 2003 Mar;10(3):193-200. doi: 10.1038/sj.cgt.7700555. Cancer Gene Ther. 2003. PMID: 12637940
-
Targeting Interferon-α Increases Antitumor Efficacy and Reduces Hepatotoxicity of E1A-mutated Spread-enhanced Oncolytic Adenovirus.Mol Ther. 2007 Mar;15(3):598-607. doi: 10.1038/sj.mt.6300064. Epub 2016 Dec 8. Mol Ther. 2007. PMID: 28182898
-
Functions and mechanisms of action of the adenovirus E3 proteins.Int Rev Immunol. 2004 Jan-Apr;23(1-2):75-111. doi: 10.1080/08830180490265556. Int Rev Immunol. 2004. PMID: 14690856 Review.
-
Adenovirus vectors for gene therapy, vaccination and cancer gene therapy.Curr Gene Ther. 2013 Dec;13(6):421-33. doi: 10.2174/1566523213666131125095046. Curr Gene Ther. 2013. PMID: 24279313 Free PMC article. Review.
Cited by
-
Characterization of human adenovirus serotypes 5, 6, 11, and 35 as anticancer agents.Virology. 2009 Nov 25;394(2):311-20. doi: 10.1016/j.virol.2009.08.038. Epub 2009 Sep 18. Virology. 2009. PMID: 19765790 Free PMC article.
-
Highly attenuated recombinant vesicular stomatitis virus VSV-12'GFP displays immunogenic and oncolytic activity.J Virol. 2013 Jan;87(2):1019-34. doi: 10.1128/JVI.01106-12. Epub 2012 Nov 7. J Virol. 2013. PMID: 23135719 Free PMC article.
-
Current issues and future directions of oncolytic adenoviruses.Mol Ther. 2010 Feb;18(2):243-50. doi: 10.1038/mt.2009.266. Epub 2009 Nov 24. Mol Ther. 2010. PMID: 19935777 Free PMC article. Review.
-
Role of Gene Therapy in Pancreatic Cancer-A Review.Cancers (Basel). 2018 Apr 3;10(4):103. doi: 10.3390/cancers10040103. Cancers (Basel). 2018. PMID: 29614005 Free PMC article. Review.
-
Going viral with cancer immunotherapy.Nat Rev Cancer. 2014 Aug;14(8):559-67. doi: 10.1038/nrc3770. Epub 2014 Jul 3. Nat Rev Cancer. 2014. PMID: 24990523 Review.
References
-
- Kirn D. Clinical research results with dl1520 (Onyx-015), a replication-selective adenovirus for the treatment of cancer: what have we learned? Gene Therapy. 2001;8:89–98. - PubMed
-
- Hermiston T. A demand for next-generation oncolytic adenoviruses. Curr Opin Mol Ther. 2006;8:322–330. - PubMed
-
- Khuri FR, Nemunaitis J, Ganly I, Arseneau J, Tannock IF, Romel L, et al. A controlled trial of intratumoral ONYX-015, a selectively-replicating adenovirus, in combination with cisplatin and 5-fluorouracil in patients with recurrent head and neck cancer. Nat Med. 2000;6:879–885. - PubMed
-
- Garber K. China approves world’s first oncolytic virus therapy for cancer treatment. J Natl Cancer Inst. 2006;98:298–300. - PubMed
-
- Shashkova EV, Spencer JF, Wold WS, Doronin K. Targeting interferon-alpha increases antitumor efficacy and reduces hepatotoxicity of E1A-mutated spread-enhanced oncolytic adenovirus. Mol Ther. 2007;15:598–607. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

