Evaluating intramural virtual electrodes in the myocardial wedge preparation: simulations of experimental conditions
- PMID: 17993491
- PMCID: PMC2242738
- DOI: 10.1529/biophysj.107.121343
Evaluating intramural virtual electrodes in the myocardial wedge preparation: simulations of experimental conditions
Abstract
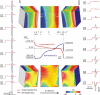
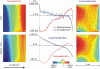
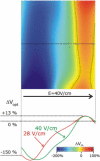
While defibrillation is the only means for prevention of sudden cardiac death, key aspects of the process, such as the intramural virtual electrodes (VEs), remain controversial. Experimental studies had attempted to assess intramural VEs by using wedge preparations and recording activity from the cut surface; however, applicability of this approach remains unclear. These studies found, surprisingly, that for strong shocks, the entire cut surface was negatively polarized, regardless of boundary conditions. The goal of this study is to examine, by means of bidomain simulations, whether VEs on the cut surface represent a good approximation to VEs in depth of the intact wall. Furthermore, we aim to explore mechanisms that could give rise to negative polarization on the cut surface. A model of wedge preparation was used, in which fiber orientation could be changed, and where the cut surface was subjected to permeable and impermeable boundary conditions. Small-scale mechanisms for polarization were also considered. To determine whether any distortions in the recorded VEs arise from averaging during optical mapping, a model of fluorescent recording was employed. The results indicate that, when an applied field is spatially uniform and impermeable boundary conditions are enforced, regardless of the fiber orientation VEs on the cut surface faithfully represent those intramurally, provided tissue properties are not altered by dissection. Results also demonstrate that VEs are sensitive to the conductive layer thickness above the cut surface. Finally, averaging during fluorescent recordings results in large negative VEs on the cut surface, but these do not arise from small-scale heterogeneities.
Figures




Similar articles
-
Do intramural virtual electrodes facilitate successful defibrillation? Model-based analysis of experimental evidence.J Cardiovasc Electrophysiol. 2006 Mar;17(3):305-11. doi: 10.1111/j.1540-8167.2006.00360.x. J Cardiovasc Electrophysiol. 2006. PMID: 16643406
-
Near-threshold field stimulation: intramural versus surface activation.Cardiovasc Res. 2006 Jan;69(1):98-106. doi: 10.1016/j.cardiores.2005.08.012. Epub 2005 Oct 13. Cardiovasc Res. 2006. PMID: 16226236
-
Virtual electrode polarization in the far field: implications for external defibrillation.Am J Physiol Heart Circ Physiol. 2000 Sep;279(3):H1055-70. doi: 10.1152/ajpheart.2000.279.3.H1055. Am J Physiol Heart Circ Physiol. 2000. PMID: 10993768
-
Imaging of Ventricular Fibrillation and Defibrillation: The Virtual Electrode Hypothesis.Adv Exp Med Biol. 2015;859:343-65. doi: 10.1007/978-3-319-17641-3_14. Adv Exp Med Biol. 2015. PMID: 26238060 Free PMC article. Review.
-
Developing a novel comprehensive framework for the investigation of cellular and whole heart electrophysiology in the in situ human heart: historical perspectives, current progress and future prospects.Prog Biophys Mol Biol. 2014 Aug;115(2-3):252-60. doi: 10.1016/j.pbiomolbio.2014.06.004. Epub 2014 Jun 24. Prog Biophys Mol Biol. 2014. PMID: 24972083 Review.
Cited by
-
Cardiac response to low-energy field pacing challenges the standard theory of defibrillation.Circ Arrhythm Electrophysiol. 2015 Jun;8(3):685-93. doi: 10.1161/CIRCEP.114.002661. Epub 2015 Mar 15. Circ Arrhythm Electrophysiol. 2015. PMID: 25772543 Free PMC article.
-
Termination of atrial fibrillation using pulsed low-energy far-field stimulation.Circulation. 2009 Aug 11;120(6):467-76. doi: 10.1161/CIRCULATIONAHA.108.825091. Epub 2009 Jul 27. Circulation. 2009. PMID: 19635972 Free PMC article.
-
Generation of histo-anatomically representative models of the individual heart: tools and application.Philos Trans A Math Phys Eng Sci. 2009 Jun 13;367(1896):2257-92. doi: 10.1098/rsta.2009.0056. Philos Trans A Math Phys Eng Sci. 2009. PMID: 19414455 Free PMC article.
-
Three-dimensional histology: tools and application to quantitative assessment of cell-type distribution in rabbit heart.Europace. 2014 Nov;16 Suppl 4(Suppl 4):iv86-iv95. doi: 10.1093/europace/euu234. Europace. 2014. PMID: 25362175 Free PMC article.
-
Representing cardiac bidomain bath-loading effects by an augmented monodomain approach: application to complex ventricular models.IEEE Trans Biomed Eng. 2011 Apr;58(4):1066-75. doi: 10.1109/TBME.2010.2096425. Epub 2011 Jan 31. IEEE Trans Biomed Eng. 2011. PMID: 21292591 Free PMC article.
References
-
- Rodriguez B., Eason J., Trayanova N. Differences between left and right ventricular anatomy determine the types of reentrant circuits induced by an external electric shock. A rabbit heart simulation study. Progr. Biophys. Mol. Biol. 2006;90:399–413. - PubMed
-
- Trayanova N. Defibrillation of the heart: insights into mechanisms from modeling studies. Exp. Physiol. 2006;91:323–337. - PubMed
-
- Roth B.J., Krassowska W. The induction of reentry in cardiac tissue. The missing link: how electric fields alter transmembrane potential. Chaos. 1998;8:204–220. - PubMed
-
- Byars J.L., Smith W.M., Ideker R.E., Fast V.G. Development of an optrode for intramural multisite optical recordings of Vm in the heart. J. Cardiovasc. Electrophysiol. 2003;14:1196–1202. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

