Conditional lethality, division defects, membrane involution, and endocytosis in mre and mrd shape mutants of Escherichia coli
- PMID: 17993535
- PMCID: PMC2258658
- DOI: 10.1128/JB.01322-07
Conditional lethality, division defects, membrane involution, and endocytosis in mre and mrd shape mutants of Escherichia coli
Abstract
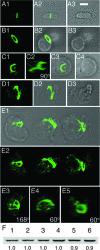
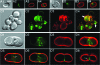
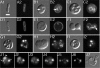
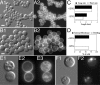
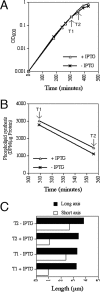
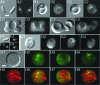
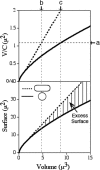
Maintenance of rod shape in Escherichia coli requires the shape proteins MreB, MreC, MreD, MrdA (PBP2), and MrdB (RodA). How loss of the Mre proteins affects E. coli viability has been unclear. We generated Mre and Mrd depletion strains under conditions that minimize selective pressure for undefined suppressors and found their phenotypes to be very similar. Cells lacking one or more of the five proteins were fully viable and propagated as small spheres under conditions of slow mass increase but formed large nondividing spheroids with noncanonical FtsZ assembly patterns at higher mass doubling rates. Extra FtsZ was sufficient to suppress lethality in each case, allowing cells to propagate as small spheres under any condition. The failure of each unsuppressed mutant to divide under nonpermissive conditions correlated with the presence of elaborate intracytoplasmic membrane-bound compartments, including vesicles/vacuoles and more-complex systems. Many, if not all, of these compartments formed by FtsZ-independent involution of the cytoplasmic membrane (CM) rather than de novo. Remarkably, while some of the compartments were still continuous with the CM and the periplasm, many were topologically separate, indicating they had been released into the cytoplasm by an endocytic-like membrane fission event. Notably, cells failed to adjust the rate of phospholipid synthesis to their new surface requirements upon depletion of MreBCD, providing a rationale for the "excess" membrane in the resulting spheroids. Both FtsZ and MinD readily assembled on intracytoplasmic membrane surfaces, and we propose that this contributes significantly to the lethal division block seen in all shape mutants under nonpermissive conditions.
Figures




References
-
- Barbour, A. G., L. W. Mayer, and B. G. Spratt. 1981. Mecillinam resistance in Escherichia coli: dissociation of growth inhibition and morphologic change. J. Infect. Dis. 143114-121. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

