Enhanced tyrosine hydroxylase expression in PC12 cells co-cultured with feline mesenchymal stem cells
- PMID: 17993752
- PMCID: PMC2868154
- DOI: 10.4142/jvs.2007.8.4.377
Enhanced tyrosine hydroxylase expression in PC12 cells co-cultured with feline mesenchymal stem cells
Abstract
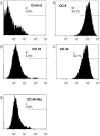
Mesenchymal stem cells (MSCs) secrete a variety of neuroregulatory molecules, such as nerve growth factor, brain-derived neurotrophic factor, and glial cell-derived neurotrophic factor, which upregulate tyrosine hydroxylase (TH) gene expression in PC12 cells. Enhancing TH gene expression is a critical step for treatment of Parkinson's disease (PD). The objective of this study was to assess the effects of co-culturing PC12 cells with MSCs from feline bone marrow on TH protein expression. We divided the study into three groups: an MSC group, a PC12 cell group, and the combined MSC + PC12 cell group (the co-culture group). All cells were cultured in DMEM-HG medium supplemented with 10% fetal bovine serum for three days. Thereafter, the cells were examined using western blot analysis and immunocytochemistry. In western blots, the co-culture group demonstrated a stronger signal at 60 kDa than the PC12 cell group (p < 0.001). TH was not expressed in the MSC group, either in western blot or immunocytochemistry. Thus, the MSCs of feline bone marrow can up-regulate TH expression in PC12 cells. This implies a new role for MSCs in the neurodegenerative disease process.
Figures


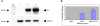

References
-
- Abouelfetouh A, Kondoh T, Ehara K, Kohmura E. Morphological differentiation of bone marrow stromal cells into neuron-like cells after co-culture with hippocampal slice. Brain Res. 2004;1029:114–119. - PubMed
-
- Arts MP, Cools AR. 6-hydroxydopamine lesion in the A8 cell group of cats produces a short-lasting decreased accuracy in goal-directed forepaw-movements. Behav Brain Res. 1999;103:13–21. - PubMed
-
- Badoyannis HC, Sharma SC, Sabban EL. The differential effects of cell density and NGF on the expression of tyrosine hydroxylase and dopamine beta-hydroxylase in PC12 cells. Brain Res Mol Brain Res. 1991;11:79–87. - PubMed
-
- Crigler L, Robey RC, Asawachaicharn A, Gaupp D, Phinney DG. Human mesenchymal stem cell subpopulations express a variety of neuro-regulatory molecules and promote neuronal cell survival and neuritogenesis. Exp Neurol. 2006;198:54–64. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

