PLGA nanoparticle--peptide conjugate effectively targets intercellular cell-adhesion molecule-1
- PMID: 17997512
- PMCID: PMC2535939
- DOI: 10.1021/bc700227z
PLGA nanoparticle--peptide conjugate effectively targets intercellular cell-adhesion molecule-1
Abstract
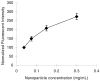
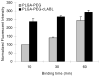
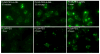
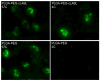
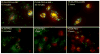
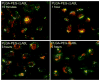
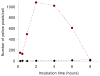
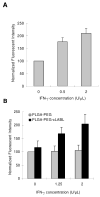
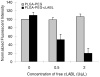
Targeted delivery of therapeutics possesses the potential to localize therapeutic agents to a specific tissue as a mechanism to enhance treatment efficacy and abrogate side effects. Antibodies have been used clinically as therapeutic agents and are currently being explored for targeting drug-loaded nanoparticles. Peptides such as RGD peptides are also being developed as an inexpensive and stable alternative to antibodies. In this study, cyclo(1,12)PenITDGEATDSGC (cLABL) peptide was used to target nanoparticles to human umbilical cord vascular endothelial cell (HUVEC) monolayers that have upregulated intercellular cell-adhesion molecule-1 (ICAM-1) expression. The cLABL peptide has been previously demonstrated to possess high avidity for ICAM-1 receptors on the cell surface. Poly( dl-lactic-coglycolic acid) nanoparticles conjugated with polyethylene glycol and cLABL demonstrated rapid binding to HUVEC with upregulated ICAM-1, which was induced by treating cells with the proinflammatory cytokine, interferon-gamma. Binding of the nanoparticles could be efficiently blocked by preincubating cells with free peptide suggesting that the binding of the nanoparticles is specifically mediated by surface peptide binding to ICAM-1 on HUVEC. The targeted nanoparticles were rapidly endocytosed and trafficked to lysosomes to a greater extent than the untargeted PLGA-PEG nanoparticles. Verification of peptide-mediated nanoparticle targeting to ICAM-1 may ultimately lead to targeting therapeutic agents to inflammatory sites expressing upregulated ICAM-1.
Figures

Similar articles
-
Controlling ligand surface density optimizes nanoparticle binding to ICAM-1.J Pharm Sci. 2011 Mar;100(3):1045-56. doi: 10.1002/jps.22342. Epub 2010 Oct 4. J Pharm Sci. 2011. PMID: 20922813 Free PMC article.
-
ICAM-1 targeting of doxorubicin-loaded PLGA nanoparticles to lung epithelial cells.Eur J Pharm Sci. 2009 May 12;37(2):141-50. doi: 10.1016/j.ejps.2009.02.008. Epub 2009 Feb 27. Eur J Pharm Sci. 2009. PMID: 19429421 Free PMC article.
-
Endothelial targeting of high-affinity multivalent polymer nanocarriers directed to intercellular adhesion molecule 1.J Pharmacol Exp Ther. 2006 Jun;317(3):1161-9. doi: 10.1124/jpet.105.098970. Epub 2006 Feb 27. J Pharmacol Exp Ther. 2006. PMID: 16505161
-
Potential of mZD7349-conjugated PLGA nanoparticles for selective targeting of vascular cell-adhesion molecule-1 in inflamed endothelium.Microvasc Res. 2016 Jul;106:110-6. doi: 10.1016/j.mvr.2016.04.003. Epub 2016 Apr 20. Microvasc Res. 2016. PMID: 27105996
-
Pegylated poly(lactide) and poly(lactide-co-glycolide) nanoparticles: preparation, properties and possible applications in drug delivery.Curr Drug Deliv. 2004 Oct;1(4):321-33. doi: 10.2174/1567201043334605. Curr Drug Deliv. 2004. PMID: 16305394 Review.
Cited by
-
Assessment of CafA Targeted BAR-Encapsulated Nanoparticles against Oral Biofilms.Pharmaceutics. 2020 Sep 1;12(9):835. doi: 10.3390/pharmaceutics12090835. Pharmaceutics. 2020. PMID: 32882864 Free PMC article.
-
Controlling ligand surface density optimizes nanoparticle binding to ICAM-1.J Pharm Sci. 2011 Mar;100(3):1045-56. doi: 10.1002/jps.22342. Epub 2010 Oct 4. J Pharm Sci. 2011. PMID: 20922813 Free PMC article.
-
Surface-modified nanotherapeutics targeting atherosclerosis.Biomater Sci. 2022 Sep 27;10(19):5459-5471. doi: 10.1039/d2bm00660j. Biomater Sci. 2022. PMID: 35980230 Free PMC article. Review.
-
Vascular Nanomedicine: Current Status, Opportunities, and Challenges.Semin Thromb Hemost. 2020 Jul;46(5):524-544. doi: 10.1055/s-0039-1692395. Epub 2019 Jun 14. Semin Thromb Hemost. 2020. PMID: 31200393 Free PMC article. Review.
-
Enhanced intracellular delivery and controlled drug release of magnetic PLGA nanoparticles modified with transferrin.Acta Pharmacol Sin. 2017 Jun;38(6):943-953. doi: 10.1038/aps.2017.45. Epub 2017 May 29. Acta Pharmacol Sin. 2017. PMID: 28552909 Free PMC article.
References
-
- Schulze-Koops H, Burkhardt H, Kalden JR. What we have learned from trials of immunomodulatory agents in rheumatoid arthritis: Future directions. Drugs Today (Barc) 1999;35:327–51. - PubMed
-
- Eniola AO, Hammer DA. Characterization of biodegradable drug delivery vehicles with the adhesive properties of leukocytes II: effect of degradation on targeting activity. Biomaterials. 2005;26:661–70. - PubMed
-
- Omolola Eniola A, Hammer DA. In vitro characterization of leukocyte mimetic for targeting therapeutics to the endothelium using two receptors. Biomaterials. 2005;26:7136–44. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

