Patients with type 2 diabetes inadequately controlled on premixed insulin: effect of initiating insulin glargine plus oral antidiabetic agents on glycaemic control in daily practice
- PMID: 17997807
- PMCID: PMC2228388
- DOI: 10.1111/j.1742-1241.2007.01598.x
Patients with type 2 diabetes inadequately controlled on premixed insulin: effect of initiating insulin glargine plus oral antidiabetic agents on glycaemic control in daily practice
Abstract
Aim: Premixed insulin regimens are commonly used for type 2 diabetes mellitus (T2DM) patients. However, there is limited information regarding next-step therapy options in cases where premixed insulin does not provide adequate glycaemic control. This 12-week observational study of everyday clinical practice evaluated the efficacy and safety of insulin glargine (glargine) plus oral antidiabetic drugs (OADs) in T2DM patients previously treated with premixed insulin.
Methods: Type 2 diabetes mellitus patients taking premixed insulin were identified from German clinics and were eligible to switch to glargine plus OADs at the physicians' and patients' discretion, as part of routine clinical practice. The study design and conduct was in accordance with German regulations. Fasting blood glucose (FBG), 2-h postprandial blood glucose (PPBG) and glycosylated haemoglobin (HbA(1c)) were measured at the start and after a 12-week observation period.
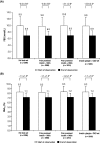
Results: A total of 5045 patients were followed-up and received glargine plus OADs. FBG [start to end-point: 9.9 +/- 2.7 to 6.9 +/- 1.5 mmol/l (178 +/- 48 to 124 +/- 26 mg/dl); p < or = 0.001], 2-h PPBG [10.8 +/- 2.8 to 7.8 +/- 1.5 mmol/l (195 +/- 50 to 140 +/- 27 mg/dl)] and HbA(1c) (8.3 +/- 1.2 to 7.2 +/- 0.8%; p < or = 0.001) improved significantly from start to end-point, respectively. A total of 48.9%, 38.4% and 73.9% of patients had FBG < 6.7 mmol/l (< 120 mg/dl), 2-h PPBG < 7.2 mmol/l (< 130 mg/dl) or HbA(1c) < 7.5%, respectively, after 12 weeks. Significant reductions in body weight were observed between the start and end of the observation period. A total of 71 adverse events were reported by 38 patients. Hypoglycaemia was the most common event (n = 16).
Conclusions: This observational study shows that, in T2DM patients inadequately controlled with premixed insulin, switching therapy to glargine plus OADs is associated with significant improvements in FBG and HbA(1c), and is well tolerated in everyday clinical practice. Further intensification of insulin therapy, perhaps by adding one or more injections of prandial insulin, would help provide further improvements in glycaemic control in these patients.
Figures
Similar articles
-
Comparison of basal insulin added to oral agents versus twice-daily premixed insulin as initial insulin therapy for type 2 diabetes.Diabetes Care. 2005 Feb;28(2):254-9. doi: 10.2337/diacare.28.2.254. Diabetes Care. 2005. PMID: 15677775 Clinical Trial.
-
Combination of oral antidiabetic agents with basal insulin versus premixed insulin alone in randomized elderly patients with type 2 diabetes mellitus.J Am Geriatr Soc. 2007 Feb;55(2):182-8. doi: 10.1111/j.1532-5415.2007.01043.x. J Am Geriatr Soc. 2007. PMID: 17302653 Clinical Trial.
-
Improved glycaemic control with BIAsp 30 in insulin-naïve type 2 diabetes patients inadequately controlled on oral antidiabetics: subgroup analysis from the IMPROVE study.Curr Med Res Opin. 2009 Nov;25(11):2643-54. doi: 10.1185/03007990903276745. Curr Med Res Opin. 2009. PMID: 19751116
-
Repaglinide : a pharmacoeconomic review of its use in type 2 diabetes mellitus.Pharmacoeconomics. 2004;22(6):389-411. doi: 10.2165/00019053-200422060-00005. Pharmacoeconomics. 2004. PMID: 15099124 Review.
-
Options for the intensification of insulin therapy when basal insulin is not enough in type 2 diabetes mellitus.Diabetes Obes Metab. 2008 Jul;10 Suppl 2:76-82. doi: 10.1111/j.1463-1326.2008.00846.x. Diabetes Obes Metab. 2008. PMID: 18577159 Review.
Cited by
-
Clinical Evidence and Practice-Based Guidelines on the Utility of Basal Insulin Combined Oral Therapy (Metformin and Glimepiride) in the Current Era.Curr Diabetes Rev. 2023;19(8):e090123212444. doi: 10.2174/1573399819666230109104300. Curr Diabetes Rev. 2023. PMID: 36624650 Free PMC article. Review.
-
Real world outcomes of adding rapid-acting insulin versus switching to analog premix insulin among US patients with type 2 diabetes treated with insulin glargine.Patient Prefer Adherence. 2013 Sep 19;7:951-60. doi: 10.2147/PPA.S49287. eCollection 2013. Patient Prefer Adherence. 2013. PMID: 24086105 Free PMC article.
-
Switching from premixed insulin to glargine-based insulin regimen improves glycaemic control in patients with type 1 or type 2 diabetes: a retrospective primary-care-based analysis.Cardiovasc Diabetol. 2009 Feb 16;8:9. doi: 10.1186/1475-2840-8-9. Cardiovasc Diabetol. 2009. PMID: 19220880 Free PMC article.
-
Starting insulin in type 2 diabetes: Overcoming barriers to insulin therapy.Int J Diabetes Dev Ctries. 2008 Apr;28(2):65-8. doi: 10.4103/0973-3930.43102. Int J Diabetes Dev Ctries. 2008. PMID: 19902051 Free PMC article.
-
Improved glycaemic control by switching from insulin NPH to insulin glargine: a retrospective observational study.Cardiovasc Diabetol. 2009 Jan 19;8:3. doi: 10.1186/1475-2840-8-3. Cardiovasc Diabetol. 2009. PMID: 19152692 Free PMC article.
References
-
- Koivisto VA, Tuominen JA, Ebeling P. Lispro Mix25 insulin as premeal therapy in type 2 diabetic patients. Diabetes Care. 1999;22:459–62. - PubMed
-
- Garber AJ. Premixed insulin analogues for the treatment of diabetes mellitus. Drugs. 2006;66:31–49. - PubMed
-
- Roach P, Trautmann M, Arora V, et al. Improved postprandial blood glucose control and reduced nocturnal hypoglycemia during treatment with two novel insulin lispro-protamine formulations, insulin lispro mix25 and insulin lispro mix50. Mix50 Study Group. Clin Ther. 1999;21:523–34. - PubMed
-
- Janka HU, Plewe G, Riddle MC, et al. Comparison of basal insulin added to oral agents versus twice-daily premixed insulin as initial insulin therapy for type 2 diabetes. Diabetes Care. 2005;28:254–9. - PubMed
-
- sanofi-aventis. LANTUS: Summary of Product Characteristics. 2006. http://www.emea.europa.eu/humandocs/Humans/EPAR/lantus/lantus.htm (accessed May 2007)
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

