Visualizing flock house virus infection in Drosophila cells with correlated fluorescence and electron microscopy
- PMID: 17998167
- PMCID: PMC2408891
- DOI: 10.1016/j.jsb.2007.09.009
Visualizing flock house virus infection in Drosophila cells with correlated fluorescence and electron microscopy
Abstract
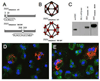
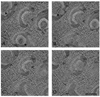
Virus assembly occurs in a complex environment and is dependent upon viral and cellular components being properly correlated in time and space. The simplicity of the flock house virus (FHV) capsid and the extensive structural, biochemical and genetic characterization of the virus make it an excellent system for studying in vivo virus assembly. The tetracysteine motif (CCPGCC), that induces fluorescence in bound biarsenical compounds (FlAsH and ReAsH), was genetically inserted in the coat protein, to visualize this gene product during virus infection. The small size of this modification when compared to those made by traditional fluorescent proteins minimizes disruption of the coat proteins numerous functions. ReAsH not only fluoresces when bound to the tetracysteine motif but also allows correlated electron microscopy (EM) of the same cell following photoconversion and osmium staining. These studies demonstrated that the coat protein was concentrated in discrete patches in the cell. High pressure freezing (HPF) followed by freeze substitution (FS) of infected cells showed that these patches were formed by virus particles in crystalline arrays. EM tomography (EMT) of the HPF/FS prepared samples showed that these arrays were proximal to highly modified mitochondria previously established to be the site of RNA replication. Two features of the mitochondrial modification are approximately 60 nm spherules that line the outer membrane and the large chamber created by the convolution induced in the entire organelle.
Figures


References
-
- Adams SR, Campbell RE, Gross LA, Martin BR, Walkup GK, Yao Y, Llopis J, Tsien RY. New biarsenical ligands and tetracysteine motifs for protein labeling in vitro and in vivo: synthesis and biological applications. Journal of the American Chemical Society. 2002;124:6063–6076. - PubMed
-
- Fisher AJ, Johnson JE. Ordered duplex RNA controls capsid architecture in an icosahedral animal virus. Nature. 1993;361:176–179. - PubMed
-
- Gaietta G, Deerinck TJ, Adams SR, Bouwer J, Tour O, Laird DW, Sosinsky GE, Tsien RY, Ellisman MH. Multicolor and electron microscopic imaging of connexin trafficking. Science. 2002;296:503–507. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

