Theoretical basis of hemodynamic MR imaging techniques to measure cerebral blood volume, cerebral blood flow, and permeability
- PMID: 17998415
- PMCID: PMC8134263
- DOI: 10.3174/ajnr.A0831
Theoretical basis of hemodynamic MR imaging techniques to measure cerebral blood volume, cerebral blood flow, and permeability
Abstract
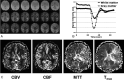
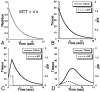
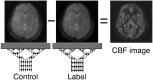
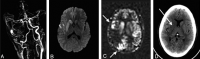
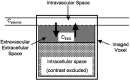
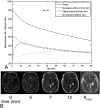
Cerebrovascular hemodynamic assessment adds new information to standard anatomic MR imaging and improves patient care. This article reviews the theoretic underpinnings of several potentially quantitative MR imaging-based methods that shed light on the hemodynamic status of the brain, including cerebral blood flow (CBF), cerebral blood volume (CBV), and contrast agent permeability. Techniques addressed include dynamic susceptibility contrast (which most simply and accurately estimates CBV), arterial spin labeling (a powerful method to measure CBF), and contrast-enhanced methods to derive permeability parameters such as the transport constant Ktrans.
Figures
References
-
- Fisel CR, Ackerman JL, Buxton RB, et al. MR contrast due to microscopically heterogeneous magnetic susceptibility: numerical simulations and applications to cerebral physiology. Magn Reson Med 1991;17:336–47 - PubMed
-
- Kiselev VG. On the theoretical basis of perfusion measurements by dynamic susceptibility contrast MRI. Magn Reson Med 2001;46:1113–22 - PubMed
-
- Donahue KM, Krouwer HG, Rand SD, et al. Utility of simultaneously acquired gradient-echo and spin-echo cerebral blood volume and morphology maps in brain tumor patients. Magn Reson Med 2000;43:845–53 - PubMed
-
- Miyati T, Banno T, Mase M, et al. Dual dynamic contrast-enhanced MR imaging. J Magn Reson Imaging 1997;7:230–35 - PubMed
-
- Barbier EL, den Boer JA, Peters AR, et al. A model of the dual effect of gadopentetate dimeglumine on dynamic brain MR images. J Magn Reson Imaging 1999;10:242–53 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
