Common variation in the ABO glycosyltransferase is associated with susceptibility to severe Plasmodium falciparum malaria
- PMID: 18003641
- PMCID: PMC2657867
- DOI: 10.1093/hmg/ddm331
Common variation in the ABO glycosyltransferase is associated with susceptibility to severe Plasmodium falciparum malaria
Abstract
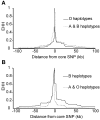
There is growing epidemiological and molecular evidence that ABO blood group affects host susceptibility to severe Plasmodium falciparum infection. The high frequency of common ABO alleles means that even modest differences in susceptibility could have a significant impact on the health of people living in malaria endemic regions. We performed an association study, the first to utilize key molecular genetic variation underlying the ABO system, genotyping >9000 individuals across three African populations. Using population- and family-based tests, we demonstrated that alleles producing functional ABO enzymes are associated with greater risk of severe malaria phenotypes (particularly malarial anemia) in comparison with the frameshift deletion underlying blood group O: case-control allelic odds ratio (OR), 1.2; 95% confidence interval (CI), 1.09-1.32; P = 0.0003; family-studies allelic OR, 1.19; 95% CI, 1.08-1.32; P = 0.001; pooled across all studies allelic OR, 1.18; 95% CI, 1.11-1.26; P = 2 x 10(-7). We found suggestive evidence of a parent-of-origin effect at the ABO locus by analyzing the family trios. Non-O haplotypes inherited from mothers, but not fathers, are significantly associated with severe malaria (likelihood ratio test of Weinberg, P = 0.046). Finally, we used HapMap data to demonstrate a region of low F(ST) (-0.001) between the three main HapMap population groups across the ABO locus, an outlier in the empirical distribution of F(ST) across chromosome 9 (approximately 99.5-99.9th centile). This low F(ST) region may be a signal of long-standing balancing selection at the ABO locus, caused by multiple infectious pathogens including P. falciparum.
Figures


References
-
- Beiguelman B, Alves FP, Moura MM, Engracia V, Nunes AC, Heckmann MI, Ferreira RG, da Silva LH, Camargo EP, Krieger H. The association of genetic markers and malaria infection in the Brazilian Western Amazonian region. Mem. Inst. Oswaldo. Cruz. 2003;98:455–460. - PubMed
-
- Fischer PR, Boone P. Short report: severe malaria associated with blood group. Am. J. Trop. Med. Hyg. 1998;58:122–123. - PubMed
-
- Lell B, May J, Schmidt-Ott RJ, Lehman LG, Luckner D, Greve B, Matousek P, Schmid D, Herbich K, Mockenhaupt FP, et al. The role of red blood cell polymorphisms in resistance and susceptibility to malaria. Clin. Infect. Dis. 1999;28:794–799. - PubMed
-
- Migot-Nabias F, Mombo LE, Luty AJ, Dubois B, Nabias R, Bisseye C, Millet P, Lu CY, Deloron P. Human genetic factors related to susceptibility to mild malaria in Gabon. Genes Immun. 2000;1:435–441. - PubMed
-
- Pant CS, Gupta DK, Sharma RC, Gautam AS, Bhatt RM. Frequency of ABO blood groups, sickle-cell haemoglobin, G-6-PD deficiency and their relation with malaria in scheduled castes and scheduled tribes of Kheda District, Gujarat. Indian J. Malariol. 1992;29:235–239. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

