Heparin enhances the furin cleavage of HIV-1 gp160 peptides
- PMID: 18037384
- PMCID: PMC7125586
- DOI: 10.1016/j.febslet.2007.11.050
Heparin enhances the furin cleavage of HIV-1 gp160 peptides
Abstract
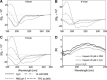
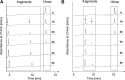
Infectious HIV-1 requires gp160 cleavage by furin at the REKR511 downward arrow motif (site1) into the gp120/gp41 complex, whereas the KAKR503 (site2) sequence remains uncleaved. We synthesized 41mer and 51mer peptides, comprising site1 and site2, to study their conformation and in vitro furin processing. We found that, while the previously reported 19mer and 13mer analogues represent excellent in vitro furin substrates, the present extended sequences require heparin for optimal processing. Our data support the hypothesis of a direct binding of heparin with site1 and site2, allowing selective exposure/accessibility of the REKR sequence, which is only then optimally cleaved by furin.
Figures



Similar articles
-
Structural investigation of the HIV-1 envelope glycoprotein gp160 cleavage site, 2: relevance of an N-terminal helix.Chembiochem. 2003 Aug 4;4(8):727-33. doi: 10.1002/cbic.200200541. Chembiochem. 2003. PMID: 12898623
-
Structural investigation and kinetic characterization of potential cleavage sites of HIV GP160 by human furin and PC1.Biochem Biophys Res Commun. 1995 Aug 4;213(1):356-61. doi: 10.1006/bbrc.1995.2137. Biochem Biophys Res Commun. 1995. PMID: 7639757
-
The disulfide loop of gp41 is critical to the furin recognition site of HIV gp160.Protein Sci. 2007 Jun;16(6):1236-41. doi: 10.1110/ps.072771407. Protein Sci. 2007. PMID: 17525470 Free PMC article.
-
Comparative functional role of PC7 and furin in the processing of the HIV envelope glycoprotein gp160.FEBS Lett. 1997 Mar 17;405(1):68-72. doi: 10.1016/s0014-5793(97)00156-7. FEBS Lett. 1997. PMID: 9094426
-
Maturation of HIV envelope glycoprotein precursors by cellular endoproteases.Biochim Biophys Acta. 2000 Nov 10;1469(3):121-32. doi: 10.1016/s0304-4157(00)00014-9. Biochim Biophys Acta. 2000. PMID: 11063880 Review.
Cited by
-
A Novel Two-Tag System for Monitoring Transport and Cleavage through the Classical Secretory Pathway - Adaptation to HIV Envelope Processing.PLoS One. 2013 Jun 19;8(6):e68835. doi: 10.1371/journal.pone.0068835. Print 2013. PLoS One. 2013. PMID: 23840860 Free PMC article.
-
Recombinant Sendai viruses expressing fusion proteins with two furin cleavage sites mimic the syncytial and receptor-independent infection properties of respiratory syncytial virus.J Virol. 2011 Mar;85(6):2771-80. doi: 10.1128/JVI.02065-10. Epub 2011 Jan 12. J Virol. 2011. PMID: 21228237 Free PMC article.
-
The Inhibition of Serine Proteases by Serpins Is Augmented by Negatively Charged Heparin: A Concise Review of Some Clinically Relevant Interactions.Int J Mol Sci. 2024 Feb 2;25(3):1804. doi: 10.3390/ijms25031804. Int J Mol Sci. 2024. PMID: 38339082 Free PMC article. Review.
-
SARS-CoV-2 S Mutations: A Lesson from the Viral World to Understand How Human Furin Works.Int J Mol Sci. 2023 Mar 1;24(5):4791. doi: 10.3390/ijms24054791. Int J Mol Sci. 2023. PMID: 36902222 Free PMC article.
-
Structural characteristics of Heparan sulfate required for the binding with the virus processing Enzyme Furin.Glycoconj J. 2022 Jun;39(3):315-325. doi: 10.1007/s10719-021-10018-8. Epub 2021 Oct 26. Glycoconj J. 2022. PMID: 34699015 Free PMC article.
References
-
- Lifson J.D., Feinberg M.B., Reyes G.R., Rabin L., Banapour B., Chakrabarti S., Moss B., Wong-Staal F., Steimer K.S., Engleman E.G., Induction of CD4-dependent cell fusion by the HTLV-III/LAV envelope glycoprotein. Nature, 323, (1986), 725– 728. - PubMed
-
- Feng Y., Broder C.C., Kennedy P.E., Berger E.A., HIV-1 entry cofactor: functional cDNA cloning of a seven-transmembrane, G protein-coupled receptor. Science, 272, (1996), 872– 877. - PubMed
-
- Dimitrov D.S., Fusin-a place for HIV-1 and T4 cells to meet. Nat. Med., 2, (1996), 640– 641. - PubMed
-
- Eckert D.M., Kim P.S., Mechanisms of viral membrane fusion and its inhibition. Annu. Rev. Biochem., 70, (2001), 777– 810. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

