ESE-1 is a potent repressor of type II collagen gene (COL2A1) transcription in human chondrocytes
- PMID: 18044710
- PMCID: PMC3937869
- DOI: 10.1002/jcp.21338
ESE-1 is a potent repressor of type II collagen gene (COL2A1) transcription in human chondrocytes
Abstract
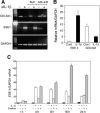
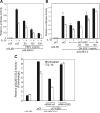
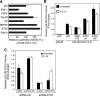
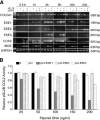
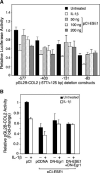
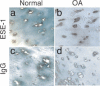
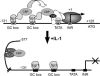
The epithelium-specific ETS (ESE)-1 transcription factor is induced in chondrocytes by interleukin-1beta (IL-1beta). We reported previously that early activation of EGR-1 by IL-1beta results in suppression of the proximal COL2A1 promoter activity by displacement of Sp1 from GC boxes. Here we report that ESE-1 is a potent transcriptional suppressor of COL2A1 promoter activity in chondrocytes and accounts for the sustained, NF-kappaB-dependent inhibition by IL-1beta. Of the ETS factors tested, this response was specific to ESE-1, since ESE-3, which was also induced by IL-1beta, suppressed COL2A1 promoter activity only weakly. In contrast, overexpression of ETS-1 increased COL2A1 promoter activity and blocked the inhibition by IL-1beta. These responses to ESE-1 and ETS-1 were confirmed using siRNA-ESE1 and siRNA-ETS1. In transient cotransfections, the inhibitory responses to ESE-1 and IL-1beta colocalized in the -577/-132 bp promoter region, ESE-1 bound specifically to tandem ETS sites at -403/-381 bp, and IL-1-induced binding of ESE-1 to the COL2A1 promoter was confirmed in vivo by ChIP. Our results indicate that ESE-1 serves a potent repressor function by interacting with at least two sites in the COL2A1 promoter. However, the endogenous response may depend upon the balance of other ETS factors such as ETS-1, and other IL-1-induced factors, including EGR-1 at any given time. Intracellular ESE-1 staining in chondrocytes in cartilage from patients with osteoarthritis (OA), but not in normal cartilage, further suggests a fundamental role for ESE-1 in cartilage degeneration and suppression of repair.
(c) 2007 Wiley-Liss, Inc.
Figures


References
-
- Andriamanalijaona R, Felisaz N, Kim SJ, King-Jones K, Lehmann M, Pujol JP, Boumediene K. Mediation of interleukin-1b-induced transforming growth factor b1 expression by activator protein 4 transcription factor in primary cultures of bovine articular chondrocytes: Possible cooperation with activator protein 1. Arthritis Rheum. 2003;48:1569–1581. - PubMed
-
- Bau B, Gebhard PM, Haag J, Knorr T, Bartnik E, Aigner T. Relative messenger RNA expression profiling of collagenases and aggrecanases in human articular chondrocytes in vivo and in vitro. Arthritis Rheum. 2002;46:2648–2657. - PubMed
-
- Brown C, Gaspar J, Pettit A, Lee R, Gu X, Wang H, Manning C, Voland C, Goldring SR, Goldring MB, Libermann TA, Gravalllese EM, Oettgen P. ESE-1 is a novel transcriptional mediator of angiopoietin-1 expression in the setting of inflammation. J Biol Chem. 2004;279:12794–12803. - PubMed
-
- Chadjichristos C, Ghayor C, Kypriotou M, Martin G, Renard E, Ala-Kokko L, Suske G, de Crombrugghe B, Pujol JP, Galera P. Sp1 and Sp3 transcription factors mediate interleukin-1 b down-regulation of human type II collagen gene expression in articular chondrocytes. J Biol Chem. 2003;278:39762–39772. - PubMed
-
- Czuwara-Ladykowska J, Shirasaki F, Jackers P, Watson DK, Trojanowska M. Fli-1 inhibits collagen type I production in dermal fibroblasts via an Sp1-dependent pathway. J Biol Chem. 2001;276:20839–20848. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

