Salmeterol/fluticasone combination in the treatment of COPD
- PMID: 18046860
- PMCID: PMC2707153
- DOI: 10.2147/copd.2006.1.3.235
Salmeterol/fluticasone combination in the treatment of COPD
Abstract
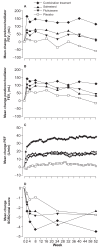
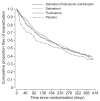
Clinical trials of a combination therapy of an inhaled corticosteroid, fluticasone propionate (FP), with a long-acting beta2-agonist, salmeterol (Sal), have demonstrated a greater improvement in lung function and in quality of life measures after the combination compared with either component of alone. In a subanalysis of the data of the TRISTAN study, Sal/FP reduced exacerbation rates in COPD patients with a baseline FEV1 < 50% of predicted. A combination therapy of budesonide and formoterol improved quality of life and FEV1, and reduced exacerbations better than either component alone. In studies of FP or of Sal/FP in COPD, there was a reduction in all-cause mortality by 25% relative to placebo. Sal/FP has anti-inflammatory effects in COPD airways. FP inhibits markers of systemic inflammation, and it is not known whether Sal/FP has an advantage over FP alone. While long-acting beta2-agonists such as Sal can be recommended for treatment of moderate COPD, addition of inhaled steroid therapy such as FP should be considered in more severe disease.
Figures
Similar articles
-
Fluticasone propionate/salmeterol 250/50 μg versus salmeterol 50 μg after chronic obstructive pulmonary disease exacerbation.Respir Res. 2014 Sep 24;15(1):105. doi: 10.1186/s12931-014-0105-2. Respir Res. 2014. PMID: 25248764 Free PMC article. Clinical Trial.
-
Glycopyrronium once-daily significantly improves lung function and health status when combined with salmeterol/fluticasone in patients with COPD: the GLISTEN study, a randomised controlled trial.Thorax. 2015 Jun;70(6):519-27. doi: 10.1136/thoraxjnl-2014-206670. Epub 2015 Apr 3. Thorax. 2015. PMID: 25841237 Free PMC article. Clinical Trial.
-
Bronchodilator effect of an inhaled combination therapy with salmeterol + fluticasone and formoterol + budesonide in patients with COPD.Respir Med. 2003 May;97(5):453-7. doi: 10.1053/rmed.2002.1455. Respir Med. 2003. PMID: 12735659 Clinical Trial.
-
Inhaled salmeterol/fluticasone propionate: a review of its use in chronic obstructive pulmonary disease.Drugs. 2004;64(17):1975-96. doi: 10.2165/00003495-200464170-00014. Drugs. 2004. PMID: 15329047 Review.
-
The role of fluticasone propionate/salmeterol combination therapy in preventing exacerbations of COPD.Int J Chron Obstruct Pulmon Dis. 2010 Jun 3;5:165-78. doi: 10.2147/copd.s4159. Int J Chron Obstruct Pulmon Dis. 2010. PMID: 20631816 Free PMC article. Review.
Cited by
-
Efficacy of salmeterol and magnesium isoglycyrrhizinate combination treatment in rats with chronic obstructive pulmonary disease.Sci Rep. 2022 Jul 19;12(1):12334. doi: 10.1038/s41598-022-16775-2. Sci Rep. 2022. PMID: 35853985 Free PMC article.
-
Relationship between the anti-inflammatory properties of salmeterol/fluticasone and the expression of CD4⁺CD25⁺Foxp3⁺ regulatory T cells in COPD.Respir Res. 2011 Oct 28;12(1):142. doi: 10.1186/1465-9921-12-142. Respir Res. 2011. PMID: 22032685 Free PMC article. Clinical Trial.
-
Chemocentric Informatics Analysis: Dexamethasone Versus Combination Therapy for COVID-19.ACS Omega. 2020 Nov 12;5(46):29765-29779. doi: 10.1021/acsomega.0c03597. eCollection 2020 Nov 24. ACS Omega. 2020. PMID: 33251412 Free PMC article.
-
Increasing Quality and Quantity of Life in Individuals with Chronic Obstructive Pulmonary Disease: A Narrative Review with an Emphasis on Pulmonary Rehabilitation.Life (Basel). 2025 May 7;15(5):750. doi: 10.3390/life15050750. Life (Basel). 2025. PMID: 40430178 Free PMC article. Review.
-
Role of the fixed combination of fluticasone and salmeterol in adult Chinese patients with asthma and COPD.Int J Chron Obstruct Pulmon Dis. 2015 Apr 15;10:775-89. doi: 10.2147/COPD.S80656. eCollection 2015. Int J Chron Obstruct Pulmon Dis. 2015. PMID: 25926729 Free PMC article. Review.
References
-
- Adcock IM, Chung KF. Why are corticosteroids ineffective in COPD? Curr Opin Investig Drugs. 2002;3:58–60. - PubMed
-
- Alsaeedi A, Sin DD, McAlister FA. The effects of inhaled corticosteroids in chronic obstructive pulmonary disease: a systematic review of randomized placebo-controlled trials. Am J Med. 2002;113:59–65. - PubMed
-
- Appleton S, Smith B, Veale A, et al. Long-acting beta2-agonists for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2000;2:CD001104. - PubMed
-
- Barnes NC, Qiu Y-S, Pavord I, et al. Anti-inflammatory effects of sameterol/fluticasone propionate in chronic obstructive lung disease. Amer J Respir Crit Care Med 2006. 2006;173:736–743. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

