Anecortave acetate in the treatment of age-related macular degeneration
- PMID: 18046876
- PMCID: PMC2695182
- DOI: 10.2147/ciia.2006.1.3.237
Anecortave acetate in the treatment of age-related macular degeneration
Abstract
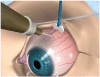
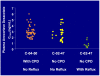
RETAANE 15mg (anecortave acetate suspension) is under investigation to treat exudative age-related macular degeneration (AMD), the single largest cause of blindness in the Western world, affecting over 15 million people in the USA. RETAANE suspension is a unique synthetic cortisene and has antiangiogenic properties that were established in multiple experimental models of angiogenesis. The molecule acts at multiple sites of the angiogenic cascade. Clinical trials in patients with exudative AMD have demonstrated the excellent safety record of both the drug anecortave acetate and the posterior juxtascleral depot (PJD) administration procedure. A pivotal study comparing RETAANE suspension with placebo showed a significantly higher chance of maintaining vision in the treatment (73%) as compared with placebo (47%). Another study compared RETAANE suspension with Visudyne photodynamic therapy, revealing no statistically significant differences between the two treatments over 24 months. AMD is a multi-faceted disease and therefore a molecule such as RETAANE suspension with a unique mechanism of action, demonstrated clinical efficacy, and retreatment every six months is an important potential treatment option which should be further investigated both as a monotherapy or in combination with other treatment strategies.
Figures


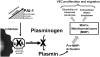
Similar articles
-
Anecortave acetate for the treatment of subfoveal choroidal neovascularization secondary to age-related macular degeneration.Eur J Ophthalmol. 2005 Jul-Aug;15(4):482-5. doi: 10.1177/112067210501500411. Eur J Ophthalmol. 2005. PMID: 16001382 Clinical Trial.
-
Anecortave acetate for the treatment of exudative age-related macular degeneration--a review of clinical outcomes.Surv Ophthalmol. 2007 Jan;52 Suppl 1:S79-90. doi: 10.1016/j.survophthal.2006.11.005. Surv Ophthalmol. 2007. PMID: 17240260 Review.
-
Safety of posterior juxtascleral depot administration of the angiostatic cortisene anecortave acetate for treatment of subfoveal choroidal neovascularization in patients with age-related macular degeneration.Graefes Arch Clin Exp Ophthalmol. 2005 Jan;243(1):9-12. doi: 10.1007/s00417-004-0961-4. Epub 2004 Jul 28. Graefes Arch Clin Exp Ophthalmol. 2005. PMID: 15290154 Clinical Trial.
-
Clinical safety profile of posterior juxtascleral depot administration of anecortave acetate 15 mg suspension as primary therapy or adjunctive therapy with photodynamic therapy for treatment of wet age-related macular degeneration.Surv Ophthalmol. 2007 Jan;52 Suppl 1:S70-8. doi: 10.1016/j.survophthal.2006.11.001. Surv Ophthalmol. 2007. PMID: 17240259 Review.
-
First clinical experience with anecortave acetate (Retaane).Klin Monbl Augenheilkd. 2007 Apr;224(4):279-81. doi: 10.1055/s-2007-962848. Klin Monbl Augenheilkd. 2007. PMID: 17458792 Clinical Trial.
Cited by
-
Leveraging Nuclear Receptors as Targets for Pathological Ocular Vascular Diseases.Int J Mol Sci. 2020 Apr 21;21(8):2889. doi: 10.3390/ijms21082889. Int J Mol Sci. 2020. PMID: 32326149 Free PMC article. Review.
-
Recent Patents on Emerging Therapeutics for the Treatment of Glaucoma, Age Related Macular Degeneration and Uveitis.Recent Pat Biomed Eng. 2012 Apr 1;5(1):83-101. doi: 10.2174/1874764711205010083. Recent Pat Biomed Eng. 2012. PMID: 25414810 Free PMC article.
-
A comprehensive insight on ocular pharmacokinetics.Drug Deliv Transl Res. 2016 Dec;6(6):735-754. doi: 10.1007/s13346-016-0339-2. Drug Deliv Transl Res. 2016. PMID: 27798766 Free PMC article. Review.
References
-
- Abdelsalam A, Del Priore L, Zarbin MA. Drusen in age-related macular degeneration: pathogenesis, natural course, and laser photocoagulation-induced regression. Surv Ophthalmol. 1999;44:1–29. - PubMed
-
- Augustin AJ, D’Amico DJ, Mieler WF, et al. Safety of posterior juxtascleral depot administration of the angiostatic cortisene anecortave acetate for treatment of subfoveal choroidal neovascularization in patients with age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol. 2005;243:9–12. - PubMed
-
- Augustin AJ, Schmidt-Erfurth U. Verteporfin therapy combined with intravitreal triamcinolone in al types of choroidal neovascularization due to age related macular degeneration. Ophthalmology. 2006;11:14–22. - PubMed
-
- BenEzra D, Griffin BW, Maftzir G, et al. Topical formulations of novel angiostatic steroids inhibit rabbit corneal neovascularization. Invest Ophthalmol Vis Sci. 1997;58:1954–62. - PubMed
-
- Blei F, Wilson EL, Mignatti P, et al. Mechanism of action of angiostatic steroids: Suppression of plasminogen activator activity via stimulation of plasminogen activator inhibitor synthesis. J Cell Physiol. 1993;155:568–78. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

