Catalytic asymmetric generation of (Z)-disubstituted allylic alcohols
- PMID: 18052173
- PMCID: PMC2532596
- DOI: 10.1021/ja0762285
Catalytic asymmetric generation of (Z)-disubstituted allylic alcohols
Abstract
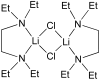
A one-pot method for the direct preparation of enantioenriched (Z)-disubstituted allylic alcohols is introduced. Hydroboration of 1-halo-1-alkynes with dicyclohexylborane, reaction with t-BuLi, and transmetalation with dialkylzinc reagents generate (Z)-disubstituted vinylzinc intermediates. In situ reaction of these reagents with aldehydes in the presence of a catalyst derived from (-)-MIB generates (Z)-disubstituted allylic alcohols. It was found that the resulting allylic alcohols were racemic, most likely due to a rapid addition reaction promoted by LiX (X = Br and Cl). To suppress the LiX-promoted reaction, a series of inhibitors were screened. It was found that 20-30 mol % tetraethylethylenediamine inhibited LiCl without inhibiting the chiral zinc-based Lewis acid. In this fashion, (Z)-disubstituted allylic alcohols were obtained with up to 98% ee. The asymmetric (Z)-vinylation could be coupled with tandem diastereoselective epoxidation reactions to provide epoxy alcohols and allylic epoxy alcohols with up to three contiguous stereogenic centers, enabling the rapid construction of complex building blocks with high levels of enantio- and diastereoselectivity.
Figures


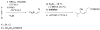



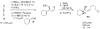
References
-
- Katsuki T, Martin VS. Org React. 1996:1.
-
- Bonini C, Righib G. Tetrahedron. 2002;58:4981–5021.
-
- Adam W, Wirth T. Acc Chem Res. 1999;32:703–710.
-
- Katsuki T. In: Comprehensive Asymmetric Catalysis. Jacobsen EN, Pfaltz A, Yamamoto H, editors. Vol. 2. Springer; Berlin: 1999. pp. 621–648.
-
- Martín VS, Woodard SS, Katsuki T, Yamada Y, Ikeda M, Sharpless KB. J Am Chem Soc. 1981;103:6237–6240.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

