Site-specific effects of tau phosphorylation on its microtubule assembly activity and self-aggregation
- PMID: 18052981
- PMCID: PMC2262108
- DOI: 10.1111/j.1460-9568.2007.05955.x
Site-specific effects of tau phosphorylation on its microtubule assembly activity and self-aggregation
Abstract
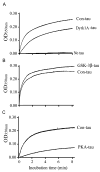
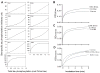
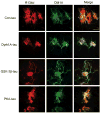
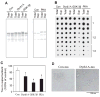
Microtubule-associated protein tau is abnormally hyperphosphorylated and aggregated into neurofibrillary tangles in brains with Alzheimer's disease. The phosphorylation sites of tau are mainly localized in the proline-rich (residues 172-251) and C-terminal tail (residues 368-441) regions, which flank the microtubule-binding repeats. Here, we investigated the effects of tau phosphorylation at these distinct sites/regions on its activity of stimulating microtubule assembly and its self-aggregation. We found that tau phosphorylation at the proline-rich region by dual-specificity tyrosine-phosphorylated and -regulated kinase 1A inhibited its microtubule assembly activity moderately and promoted its self-aggregation slightly. Tau phosphorylation at the C-terminal tail region by glycogen synthase kinase-3beta increased its activity and promoted its self-aggregation markedly. Tau phosphorylation at both regions plus the microtubule-binding region by cAMP-dependent protein kinase diminished its activity (approximately 70% inhibition) and disrupted microtubules. These studies reveal the differential regulation of tau's biological activity and self-aggregation by phosphorylation at various sites/regions.
Figures

Similar articles
-
Phosphorylation-mimicking glutamate clusters in the proline-rich region are sufficient to simulate the functional deficiencies of hyperphosphorylated tau protein.Biochem J. 2001 Aug 1;357(Pt 3):759-67. doi: 10.1042/0264-6021:3570759. Biochem J. 2001. PMID: 11463346 Free PMC article.
-
Phosphorylation that detaches tau protein from microtubules (Ser262, Ser214) also protects it against aggregation into Alzheimer paired helical filaments.Biochemistry. 1999 Mar 23;38(12):3549-58. doi: 10.1021/bi981874p. Biochemistry. 1999. PMID: 10090741
-
Phosphorylation of microtubule-associated protein tau by isoforms of c-Jun N-terminal kinase (JNK).J Neurochem. 2004 Jul;90(2):352-8. doi: 10.1111/j.1471-4159.2004.02479.x. J Neurochem. 2004. PMID: 15228592
-
Tau therapeutic strategies for the treatment of Alzheimer's disease.Curr Top Med Chem. 2006;6(6):579-95. doi: 10.2174/156802606776743057. Curr Top Med Chem. 2006. PMID: 16712493 Review.
-
Role of microtubule-associated protein tau phosphorylation in Alzheimer's disease.J Huazhong Univ Sci Technolog Med Sci. 2017 Jun;37(3):307-312. doi: 10.1007/s11596-017-1732-x. Epub 2017 Jun 6. J Huazhong Univ Sci Technolog Med Sci. 2017. PMID: 28585125 Review.
Cited by
-
Phosphorylation of tau at Thr212, Thr231, and Ser262 combined causes neurodegeneration.J Biol Chem. 2010 Oct 1;285(40):30851-60. doi: 10.1074/jbc.M110.110957. Epub 2010 Jul 27. J Biol Chem. 2010. PMID: 20663882 Free PMC article.
-
Axonal plasticity in response to active forces generated through magnetic nano-pulling.Cell Rep. 2023 Jan 31;42(1):111912. doi: 10.1016/j.celrep.2022.111912. Epub 2022 Dec 29. Cell Rep. 2023. PMID: 36640304 Free PMC article.
-
Prion and Prion-Like Protein Strains: Deciphering the Molecular Basis of Heterogeneity in Neurodegeneration.Viruses. 2019 Mar 14;11(3):261. doi: 10.3390/v11030261. Viruses. 2019. PMID: 30875755 Free PMC article. Review.
-
Leptin inhibits glycogen synthase kinase-3beta to prevent tau phosphorylation in neuronal cells.Neurosci Lett. 2009 May 22;455(3):191-4. doi: 10.1016/j.neulet.2009.03.066. Epub 2009 Mar 25. Neurosci Lett. 2009. PMID: 19429119 Free PMC article.
-
The role of DYRK1A in neurodegenerative diseases.FEBS J. 2011 Jan;278(2):236-45. doi: 10.1111/j.1742-4658.2010.07955.x. Epub 2010 Dec 13. FEBS J. 2011. PMID: 21156028 Free PMC article. Review.
References
-
- Abraha A, Ghoshal N, Gamblin TC, Cryns V, Berry RW, Kuret J, Binder LI. C-terminal inhibition of tau assembly in vitro and in Alzheimer’s disease. J Cell Sci. 2000;113:3737–3745. - PubMed
-
- Alonso A, del C, Grundke-Iqbal I, Iqbal K. Alzheimer’s disease hyperphosphorylated tau sequesters normal tau into tangles of filaments and disassembles microtubules. Nat Med. 1996;2:783–787. - PubMed
-
- Alonso A, del C, Grundke-Iqbal I, Barra HS, Iqbal K. Abnormal phosphorylation of tau and the mechanism of Alzheimer neurofibrillary degeneration: sequestration of microtubule-associated proteins 1 and 2 and the disassembly of microtubules by the abnormal tau. Proc Natl Acad Sci USA. 1997;94:298–303. - PMC - PubMed
-
- Alonso A, del C, Zaidi T, Novak M, Barra HS, Grundke-Iqbal I, Iqbal K. Interaction of tau isoforms with Alzheimer disease abnormally hyperphosphorylated tau and in vitro phosphorylation into disease like protein. J Biol Chem. 2001a;276:37 967–37 973. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

