Allergen-IgE complexes trigger CD23-dependent CCL20 release from human intestinal epithelial cells
- PMID: 18054562
- PMCID: PMC2174915
- DOI: 10.1053/j.gastro.2007.09.024
Allergen-IgE complexes trigger CD23-dependent CCL20 release from human intestinal epithelial cells
Abstract
Background & aims: In food allergic individuals, exposure to food allergens by the oral route can trigger immediate (within minutes) local hypersensitivity reactions in the intestine followed by a late-phase inflammatory response. Previous work has shown that CD23 is constitutively expressed by human intestinal epithelial cells and mediates the uptake of allergen-IgE complexes. We hypothesized that allergen-IgE complexes could also signal via CD23 to trigger an inflammatory cascade in the local environment.
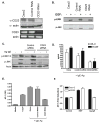
Methods: Caco-2 monolayers were stimulated with human IgE-antigen (Ag) complexes. IL-8 and CCL20 mRNA and protein were determined by RT-PCR and ELISA, respectively. Signaling pathways were assessed by immunoblotting. Endogenous CD23 expression was knocked down by stable transfection with CD23 shRNA retroviral plasmid. Migration assays were performed using human monocyte-derived dendritic cells.
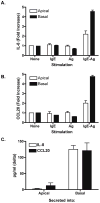
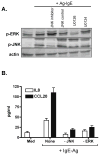
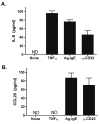
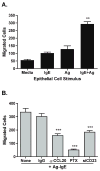
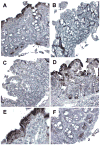
Results: Stimulation of Caco-2 cells with IgE-Ag complexes triggered upregulation of IL-8 and CCL20 at the mRNA and protein level. Allergen complexes induced phosphorylation of ERK and JNK, but not p38 MAP kinase or NK-kappaB, and resulted in AP-1 activation. Cross-linking of CD23 replicated the findings with IgE-Ag complexes, and silencing of CD23 expression abrogated the response to allergen-IgE complexes. Supernatant from IgE-Ag-stimulated epithelial cells induced migration of dendritic cells in a CCL20-dependent manner. Finally, immunostaining of duodenal biopsies demonstrated that CCL20 was constitutively expressed by epithelial cells in vivo.
Conclusions: Signaling via epithelial CD23 may participate in the late-phase inflammatory response by the release of chemokines capable of recruiting antigen presenting cells and effector cells of allergic inflammation.
Figures

Similar articles
-
Quercetin and kaempferol suppress immunoglobulin E-mediated allergic inflammation in RBL-2H3 and Caco-2 cells.Inflamm Res. 2010 Oct;59(10):847-54. doi: 10.1007/s00011-010-0196-2. Epub 2010 Apr 13. Inflamm Res. 2010. PMID: 20383790
-
Transcytosis of IgE-antigen complexes by CD23a in human intestinal epithelial cells and its role in food allergy.Gastroenterology. 2006 Jul;131(1):47-58. doi: 10.1053/j.gastro.2006.03.044. Gastroenterology. 2006. PMID: 16831589
-
CD23 provides a noninflammatory pathway for IgE-allergen complexes.J Allergy Clin Immunol. 2020 Jan;145(1):301-311.e4. doi: 10.1016/j.jaci.2019.07.045. Epub 2019 Aug 19. J Allergy Clin Immunol. 2020. PMID: 31437490
-
Consequences of IgE/CD23-mediated antigen presentation in allergy.Immunol Today. 1995 Aug;16(8):380-3. doi: 10.1016/0167-5699(95)80005-0. Immunol Today. 1995. PMID: 7546193 Review.
-
The role of allergen-specific IgE, IgG and IgA in allergic disease.Allergy. 2021 Dec;76(12):3627-3641. doi: 10.1111/all.14908. Epub 2021 Jun 8. Allergy. 2021. PMID: 33999439 Free PMC article. Review.
Cited by
-
Inhibition of CD23-mediated IgE transcytosis suppresses the initiation and development of allergic airway inflammation.Mucosal Immunol. 2015 Nov;8(6):1262-74. doi: 10.1038/mi.2015.16. Epub 2015 Mar 18. Mucosal Immunol. 2015. PMID: 25783969 Free PMC article.
-
Chronic application of alcohol-soluble gluten extract over undamaged skin causes clinical sensitization for life-threatening anaphylaxis via activation of systemic Th2 immune responses in mice.Front Allergy. 2023 Sep 29;4:1214051. doi: 10.3389/falgy.2023.1214051. eCollection 2023. Front Allergy. 2023. PMID: 37841051 Free PMC article.
-
Mucosal immunology of tolerance and allergy in the gastrointestinal tract.Immunol Res. 2012 Dec;54(1-3):75-82. doi: 10.1007/s12026-012-8308-4. Immunol Res. 2012. PMID: 22447352 Free PMC article. Review.
-
IgE regulates the expression of smMLCK in human airway smooth muscle cells.PLoS One. 2014 Apr 10;9(4):e93946. doi: 10.1371/journal.pone.0093946. eCollection 2014. PLoS One. 2014. PMID: 24722483 Free PMC article.
-
In vivo intranasal anti-CD23 treatment inhibits allergic responses in a murine model of allergic rhinitis.J Mol Histol. 2013 Jun;44(3):327-38. doi: 10.1007/s10735-013-9484-9. Epub 2013 Feb 3. J Mol Histol. 2013. PMID: 23377922
References
-
- Sicherer SH, Munoz-Furlong A, Sampson HA. Prevalence of seafood allergy in the United States determined by a random telephone survey. J Allergy Clin Immunol. 2004;114:159–65. - PubMed
-
- Sicherer SH, Munoz-Furlong A, Sampson HA. Prevalence of peanut and tree nut allergy in the United States determined by means of a random digit dial telephone survey: a 5-year follow-up study. J Allergy Clin Immunol. 2003;112:1203–7. - PubMed
-
- Lin XP, Magnusson J, Ahlstedt S, Dahlman-Hoglund A, Hanson LL, Magnusson O, Bengtsson U, Telemo E. Local allergic reaction in food-hypersensitive adults despite a lack of systemic food-specific IgE. J Allergy Clin Immunol. 2002;109:879–87. - PubMed
-
- Wershil BK, Furuta GT, Wang ZS, Galli SJ. Mast cell-dependent neutrophil and mononuclear cell recruitment in immunoglobulin E-induced gastric reactions in mice. Gastroenterology. 1996;110:1482–90. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

