The c3-v4 region is a major target of autologous neutralizing antibodies in human immunodeficiency virus type 1 subtype C infection
- PMID: 18057243
- PMCID: PMC2258729
- DOI: 10.1128/JVI.02187-07
The c3-v4 region is a major target of autologous neutralizing antibodies in human immunodeficiency virus type 1 subtype C infection
Abstract
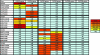
The early autologous neutralizing antibody response in human immunodeficiency virus type 1 (HIV-1) subtype C infections is often characterized by high titers, but the response is type specific with little to no cross-neutralizing activity. The specificities of these early neutralizing antibodies are not known; however, the type specificity suggests that they may target the variable regions of the envelope. Here, we show that cross-reactive anti-V3 antibodies developed within 3 to 12 weeks in six individuals but did not mediate autologous neutralization. Using a series of chimeric viruses, we found that antibodies directed at the V1V2, V4, and V5 regions contributed to autologous neutralization in some individuals, with V1V2 playing a more substantial role. However, these antibodies did not account for the total neutralizing capacity of these sera against the early autologous virus. Antibodies directed against the C3-V4 region were involved in autologous neutralization in all four sera studied. In two sera, transfer of the C3-V4 region rendered the chimera as sensitive to antibody neutralization as the parental virus. Although the C3 region, which contains the highly variable alpha2-helix was not a direct target in most cases, it contributed to the formation of neutralization epitopes as substitution of this region resulted in neutralization resistance. These data suggest that the C3 and V4 regions combine to form important structural motifs and that epitopes in this region are major targets of the early autologous neutralizing response in HIV-1 subtype C infection.
Figures





References
-
- Binley, J. M., T. Wrin, B. Korber, M. B. Zwick, M. Wang, C. Chappey, G. Stiegler, R. Kunert, S. Zolla-Pazner, H. Katinger, C. J. Petropoulos, and D. R. Burton. 2004. Comprehensive cross-clade neutralization analysis of a panel of anti-human immunodeficiency virus type 1 monoclonal antibodies. J. Virol. 7813232-13252. - PMC - PubMed
-
- Decker, J. M., F. Bibollet-Ruche, X. Wei, S. Wang, D. N. Levy, W. Wang, E. Delaporte, M. Peeters, C. A. Derdeyn, S. Allen, E. Hunter, M. S. Saag, J. A. Hoxie, B. H. Hahn, P. D. Kwong, J. E. Robinson, and G. M. Shaw. 2005. Antigenic conservation and immunogenicity of the HIV coreceptor binding site. J. Exp. Med. 2011407-1419. - PMC - PubMed
-
- Derdeyn, C. A., J. M. Decker, F. Bibollet-Ruche, J. L. Mokili, M. Muldoon, S. A. Denham, M. L. Heil, F. Kasolo, R. Musonda, B. H. Hahn, G. M. Shaw, B. T. Korber, S. Allen, and E. Hunter. 2004. Envelope-constrained neutralization-sensitive HIV-1 after heterosexual transmission. Science 3032019-2022. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

