Lipids revert inert Abeta amyloid fibrils to neurotoxic protofibrils that affect learning in mice
- PMID: 18059472
- PMCID: PMC2206134
- DOI: 10.1038/sj.emboj.7601953
Lipids revert inert Abeta amyloid fibrils to neurotoxic protofibrils that affect learning in mice
Abstract
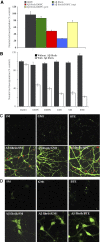
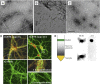
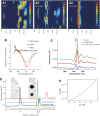
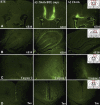
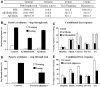
Although soluble oligomeric and protofibrillar assemblies of Abeta-amyloid peptide cause synaptotoxicity and potentially contribute to Alzheimer's disease (AD), the role of mature Abeta-fibrils in the amyloid plaques remains controversial. A widely held view in the field suggests that the fibrillization reaction proceeds 'forward' in a near-irreversible manner from the monomeric Abeta peptide through toxic protofibrillar intermediates, which subsequently mature into biologically inert amyloid fibrils that are found in plaques. Here, we show that natural lipids destabilize and rapidly resolubilize mature Abeta amyloid fibers. Interestingly, the equilibrium is not reversed toward monomeric Abeta but rather toward soluble amyloid protofibrils. We characterized these 'backward' Abeta protofibrils generated from mature Abeta fibers and compared them with previously identified 'forward' Abeta protofibrils obtained from the aggregation of fresh Abeta monomers. We find that backward protofibrils are biochemically and biophysically very similar to forward protofibrils: they consist of a wide range of molecular masses, are toxic to primary neurons and cause memory impairment and tau phosphorylation in mouse. In addition, they diffuse rapidly through the brain into areas relevant to AD. Our findings imply that amyloid plaques are potentially major sources of soluble toxic Abeta-aggregates that could readily be activated by exposure to biological lipids.
Figures
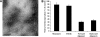
References
-
- Aksenov MY, Aksenova MV, Butterfield DA, Hensley K, Vigo-Pelfrey C, Carney JM (1996) Glutamine synthetase-induced enhancement of beta-amyloid peptide A beta (1–40) neurotoxicity accompanied by abrogation of fibril formation and A beta fragmentation. J Neurochem 66: 2050–2056 - PubMed
-
- Barghorn S, Nimmrich V, Striebinger A, Krantz C, Keller P, Janson B, Bahr M, Schmidt M, Bitner RS, Harlan J, Barlow E, Ebert U, Hillen H (2005) Globular amyloid beta-peptide oligomer—a homogenous and stable neuropathological protein in Alzheimer's disease. J Neurochem 95: 834–847 - PubMed
-
- Bitan G, Teplow DB (2004) Rapid photochemical cross-linking—a new tool for studies of metastable, amyloidogenic protein assemblies. Acc Chem Res 37: 357–364 - PubMed
-
- Bitan G, Vollers SS, Teplow DB (2003) Elucidation of primary structure elements controlling early amyloid beta-protein oligomerization. J Biol Chem 278: 34882–34889 - PubMed
-
- Booth DR, Sunde M, Bellotti V, Robinson CV, Hutchinson WL, Fraser PE, Hawkins PN, Dobson CM, Radford SE, Blake CC, Pepys MB (1997) Instability, unfolding and aggregation of human lysozyme variants underlying amyloid fibrillogenesis. Nature 385: 787–793 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

